Chinese Medical Sciences Journal ›› 2022, Vol. 37 ›› Issue (4): 293-302.doi: 10.24920/004067
显微镜下多血管炎患者继发弥漫性肺泡出血和肺间质病的临床特征及预后分析
- 中国医学科学院 北京协和医学院,北京协和医院呼吸与危重症医学科,北京 100730
-
收稿日期:2022-01-22接受日期:2022-07-12出版日期:2022-12-31发布日期:2022-08-24 -
通讯作者:施举红 E-mail:shijh@pumch.cn
Characteristics and Prognosis of Microscopic Polyangiitis Patients with Diffuse Alveolar Hemorrhage and Interstitial Lung Disease
Yu Gu,Ting Zhang,Min Peng,Juhong Shi*( )
)
- Department of Respiratory and Critical Care Medicine, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing 100037, China
-
Received:2022-01-22Accepted:2022-07-12Published:2022-12-31Online:2022-08-24 -
Contact:Juhong Shi E-mail:shijh@pumch.cn
摘要:
目的 评估继发于显微镜下多血管炎(microscopic polyangiitis,MPA)的弥漫性肺泡出血和/或肺间质病患者的临床特征以及预后相关因素。
方法 回顾性分析2002年至2012年于北京协和医院内科住院的MPA患者,根据肺受累类型不同分为单纯肺间质病组、单纯弥漫性肺泡出血组、弥漫肺泡出血合并肺间质病组和无肺受累组。比较各组患者人口学资料、全身症状、脏器受累情况、实验室检查、治疗与预后。采用Logistic回归和Cox分析患者早期与晚期死亡的危险因素。
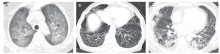
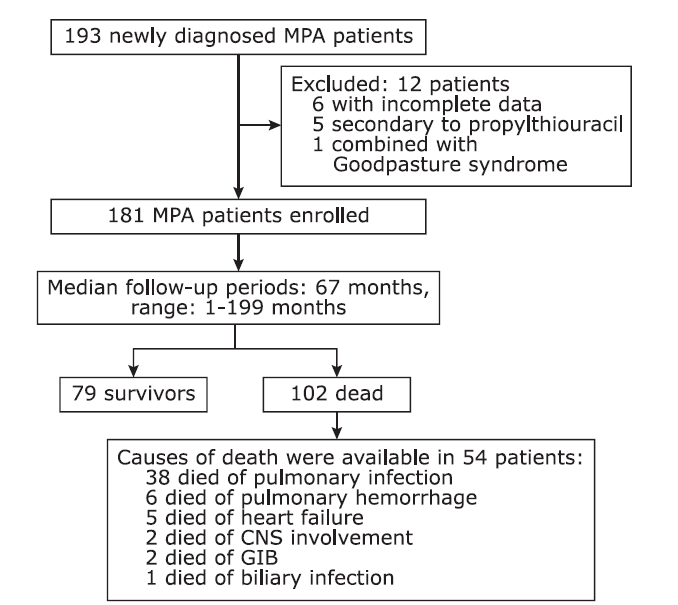
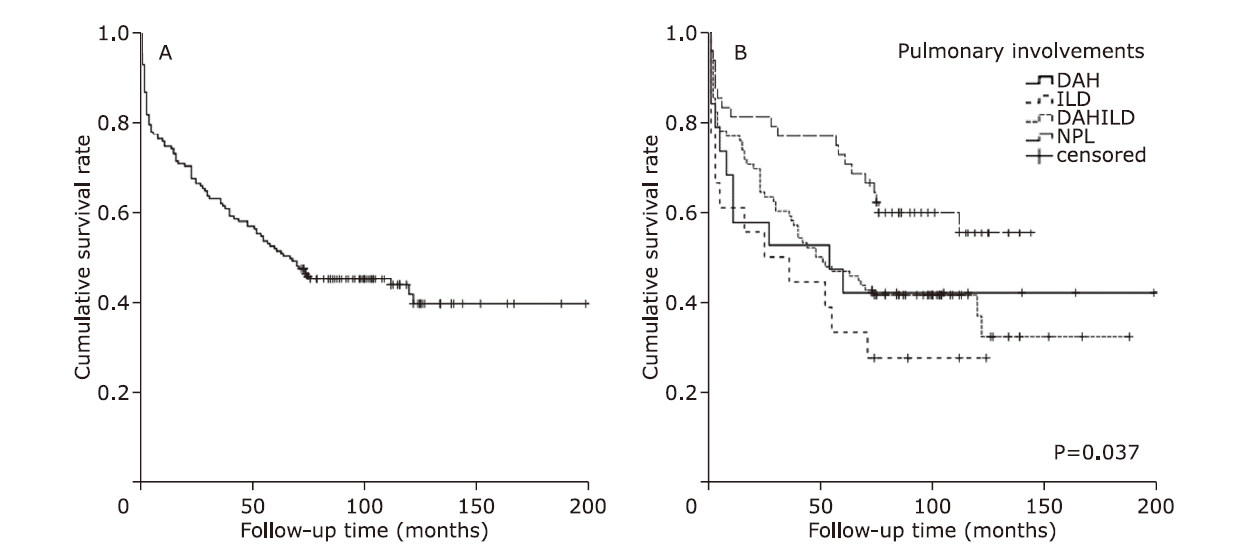
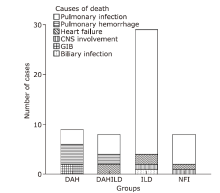
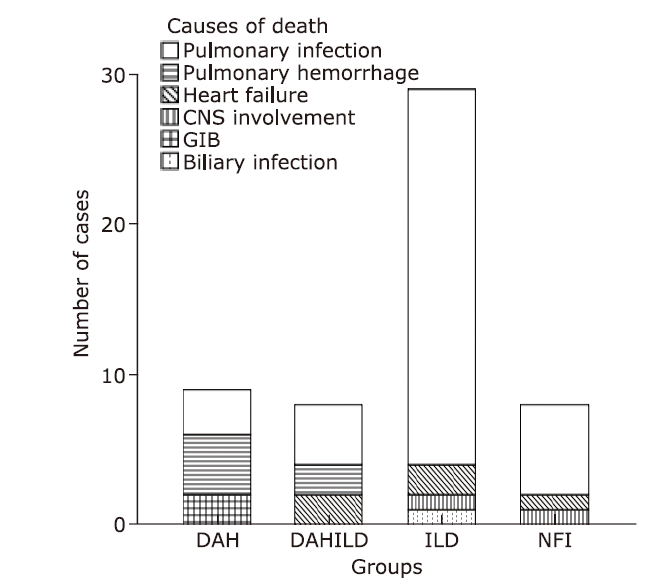
结果 共181例MPA患者纳入研究,其中单纯肺间质病组96例、单纯弥漫性肺泡出血组19例、弥漫肺泡出血合并肺间质病组18例、无肺受累组48例。随访时间的中位数为67个月(范围:1~199个月)。单纯弥漫性肺泡出血组肾脏受累最严重(血肌酐中位值:449 μmol/L,显著高于单纯肺间质病组(123 μmol/L,Nemenyi = -35.215,P = 0.045)和弥漫肺泡出血合并肺间质病组(359 μmol/L,Nemenyi = -43.609,P = 0.007)。单纯肺间质病组患者年龄大于单纯弥漫性肺泡出血组(中位年龄:69比57岁;Nemenyi = 43.853,P = 0.005)。弥漫肺泡出血合并肺间质病组患者融合了两种单纯亚型患者的临床特点,并且死亡率最高(死亡率72.2%)。MPA患者总的5年生存率为51.9%。外周血白细胞升高(1月内死亡风险评估:OR = 1.103,95%CI:1.008-1.207,P = 0.032;1年内死亡风险评估:OR = 1.103,95%CI:1.026-1.186,P = 0.008)是MPA患者早期死亡的高危因素。高龄(HR = 1.044,95%CI:1.023-1.066,P < 0.001)、肾功能严重受损(HR = 1.001,95%CI:1.000-1.002,P = 0.032)及心血管受累(HR = 2.093,95%CI:1.195-3.665,P = 0.010)是MPA患者晚期死亡的高危因素。在诊断后1年内共有49例患者发生过肺部感染。肺部感染是MPA患者最主要的死亡原因(38/54),以肺间质病患者尤为明显。
结论 不同肺受累模式的MPA患者具有完全不同的临床特征,提示不同肺受累模式的MPA可能存在不同的发病机制。因此,对继发于MPA的弥漫性肺泡出血和肺间质病应按照不同的临床亚型分别研究。
引用本文
Yu Gu, Ting Zhang, Min Peng, Juhong Shi. Characteristics and Prognosis of Microscopic Polyangiitis Patients with Diffuse Alveolar Hemorrhage and Interstitial Lung Disease[J].Chinese Medical Sciences Journal, 2022, 37(4): 293-302.
"
| Variable | DAH | DAH+ILD | ILD | NPI | Statistic | P value |
|---|---|---|---|---|---|---|
| No. of patients [n (%)] | 19 (10.5) | 18 (9.9) | 96 (53.0) | 48 (26.5) | ||
| Age at diagnosis [yrs, median (range)] | 57 (47-65)a | 64 (59-75) | 69 (60-74)a | 65 (53-74) | H = 12.13 | 0.007 |
| Male [n (%)] | 7 (36.8) | 8 (44.4) | 50 (52.1) | 20 (41.7) | χ2 = 2.378 | 0.498 |
| Follow-up time [months, median (range)] | 54 (5-105) | 31 (3-78) | 50 (15-100) | 78 (57-112) | H = 2.545 | 0.058 |
| BVAS [score, median (range)] | 22 (18-28) | 21 (12-25) | 21 (15-27) | 21 (15-29) | H = 1.955 | 0.582 |
| FFS2009 [score, mean±SD] | 2.01±0.97 | 2.22±0.81 | 1.88±0.77 | 1.81±0.76 | H = 3.356 | 0.340 |
| Fever [n (%)] | 4 (21.1) | 6 (33.3) | 25 (26.0) | 14 (29.2) | Fisher’s exact χ2=0.927 | 0.831 |
| Weight loss [n (%)] | 6 (31.6) | 9 (50.0) | 48 (50.0) | 26 (54.2) | χ2 = 2.863 | 0.413 |
| Organ involvement [n (%)] | ||||||
| Renal | 18 (94.7) | 18 (100) | 83 (86.5) | 43 (89.6) | Fisher’s exact χ2=2.951 | 0.398 |
| Nervous system | 5 (26.3) | 3 (16.7) | 40 (41.7) | 21 (48.3) | χ2 = 5.791 | 0.122 |
| ENT | 2 (10.5) | 1 (5.6) | 14 (14.6) | 12 (25.0) | Fisher’s exact χ2=4.263 | 0.235 |
| Skin | 4 (21.1) | 3 (16.7) | 10 (10.4) | 8 (16.7) | Fisher’s exact χ2=2.702 | 0.433 |
| Cardiovascular system | 4 (21.1) | 3 (16.7) | 8 (8.3) | 3 (6.3) | Fisher’s exact χ2=4.589 | 0.171 |
| Eyes | 1 (5.3) | 0 | 5 (5.2) | 5 (10.4) | Fisher’s exact χ?=2.294 | 0.467 |
| Abdomen | 3 (15.8) | 1 (5.6) | 2 (2.1) | 3 (6.3) | Fisher’s exact χ2=6.237 | 0.064 |
| Laboratory findings | ||||||
| WBC [×??9/L, median (range)] | 10.1 (8.1-18.0) | 9.4 (7.9-13.7) | 10.0 (7.4-13.7) | 9.6 (6.8-12.8) | H = 2.510 | 0.473 |
| HGB (g/L, mean±SD)* | 88±30b | 92±22 | 107±22b,c | 96±19c | F = 5.952 | 0.001 |
| PLT (×109/L, median (range)) | 291 (209-429) | 228 (132-308) | 278 (194-363) | 260 (230-405) | H = 5.387 | 0.146 |
| ESR (mm/h, mean±SD)* | 78 ± 40 | 73 ± 42 | 75±31 | 77±31 | F = 0.144 | 0.933 |
| CRP (mg/L, median (range)) | 39 (18-70) | 21 (13-73) | 45 (10-91) | 58 (14-127) | H = 0.902 | 0.825 |
| Creatinine [μmol/L, median (range)] | 449 (126-523)d,e | 357 (156-640)e | 123 (81-230)d | 170 (104-385) | H = 16.572 | 0.001 |
| ANA [n (%)] | 28 (29.2) | 7 (38.9) | 7 (36.8) | 16 (33.3) | χ2 = 0.990 | 0.825 |
| ANCA [n (%)] | Fisher’s exact χ2=10.309 | 0.185 | ||||
| PR3/c-ANCA | 5 (5.3) | 0 | 2 (2.1) | 2 (4.2) | >0.05 | |
| MPO/p-ANCA | 17 (89.5) | 16 (88.9) | 92 (95.8) | 44 (91.7) | >0.05 | |
| MPO/p-ANCA+PR3/c-ANCA positive | 0 | 2 (11.1) | 1 (1.0) | 2 (4.2) | >0.05 | |
| MPO/p-ANCA+PR3/c-ANCA negative | 1 (5.3) | 0 | 1 (1.0) | 0 | >0.05 | |
| Treatments | ||||||
| p-GC/h-GC/n-GC (n) | 8/11/0 | 8/10/0 | 20/73/3 | 17/31/0 | Fisher’s exact χ2=8.867 | 0.129 |
| iv-CTX/po-CTX/n-CTX (n) | 11/4/4 | 11/4/3 | 53/33/10 | 25/15/8 | Fisher’s exact χ2=3.848 | 0.704 |
| IVIG [n (%)] | 2 (10.5) | 5 (27.8)g | 3 (3.1)g | 3 (6.3) | Fisher’s exact χ2=11.062 | 0.005 |
| PE [n (%)] | 3 (15.8) | 5 (27.8)h | 1 (1.0)h,i | 1 (2.1)i | Fisher’s exact χ2 =18.792 | <0.001 |
| Prognosis [n (%)] | ||||||
| Pulmonary infections in one year | 4 (8.2) | 4 (8.2) | 34 (69.4) | 7 (14.3) | Fisher’s exact χ2=7.590 | 0.051 |
| Deaths in one year | 8 (42.1) | 7 (38.9) | 22 (22.9) | 9 (18.8) | Fisher’s exact χ2=5.870 | 0.115 |
| Deaths overall | 11 (57.9) | 13 (72.2) | 58 (60.4) | 20 (41.7) | χ2 = 6.715 | 0.082 |
"
| Variables | Death in one year | Death in one month | |||||
|---|---|---|---|---|---|---|---|
| OR | 95%CI | P value | OR | 95%CI | P value | ||
| Age | 1.047 | 1.009, 1.086 | 0.016 | 1.066 | 0.995, 1.142 | 0.080 | |
| Male | 0.993 | 0.465, 2.121 | 0.905 | 0.537 | 0.113, 2.333 | 0.866 | |
| Fever | 1.775 | 0.690, 4.566 | 0.302 | ||||
| DAH | 2.164 | 0.864, 5.418 | 0.078 | 4.358 | 1.277, 14.877 | 0.019 | |
| Cardiovascular system | 2.013 | 0.661, 6.129 | 0.145 | ||||
| Skin/mucosa | 5.110 | 1.392, 18.758 | 0.014 | ||||
| Abdomen | 3.744 | 0.419, 33.492 | 0.272 | ||||
| WBC | 1.103 | 1.026, 1.186 | 0.008 | 1.103 | 1.008, 1.207 | 0.032 | |
| HGB | 0.978 | 0.944, 1.014 | 0.240 | ||||
| Creatinine | 1.002 | 1.000, 1.003 | 0.011 | 1.001 | 0.998, 1.004 | 0.430 | |
"
| Variables | Cox regression | Logistic regression | |||||
|---|---|---|---|---|---|---|---|
| HR | 95%CI | P value | OR | 95%CI | P value | ||
| Age | 1.044 | 1.023, 1.066 | <0.001 | 1.061 | 1.030, 1.094 | < 0.001 | |
| Male | 0.803 | 0.539, 1.196 | 0.314 | 0.761 | 0.387, 1.497 | 0.366 | |
| ILD | 1.389 | 0.904, 2.134 | 0.145 | 1.623 | 0.792, 3.324 | 0.185 | |
| Cardiovascular system | 2.093 | 1.195, 3.665 | 0.010 | 6.216 | 1.260, 30.664 | 0.025 | |
| WBC | 1.040 | 1.002, 1.079 | 0.039 | 1.024 | 0.955, 1.098 | 0.503 | |
| HGB | 1.021 | 1.004, 1.038 | 0.016 | ||||
| Creatinine | 1.001 | 1.000, 1.002 | 0.032 | 1.003 | 1.001, 1.005 | 0.003 | |
| [1] |
Lai QY, Ma TT, Li ZY, et al. Predictors for mortality in patients with antineutrophil cytoplasmic autoantibody-associated vasculitis: a study of 398 Chinese patients. J Rheumatol 2014; 41(9):1849-55. doi: 10.3899/jrheum.131426.
doi: 10.3899/jrheum.131426 |
| [2] |
Yamagata M, Ikeda K, Tsushima K, et al. Prevalence and responsiveness to treatment of lung abnormalities on chest computed tomography in patients with microscopic polyangiitis: a multicenter, longitudinal, retrospective study of one hundred fifty consecutive hospital-based Japanese patients. Arthritis Rheumatol 2016; 68(3):713-23. doi: 10.1002/art.39475.
doi: 10.1002/art.39475 |
| [3] |
Kim MJ, Shin K. Interstitial lung disease and diffuse alveolar hemorrhage, the two key pulmonary manifestations in microscopic polyangiitis. Tuberc Respir Dis (Seoul) 2021; 84(4):255-62. doi: 10.4046/trd.2021.0065.
doi: 10.4046/trd.2021.0065 |
| [4] |
Katsumata Y, Kawaguchi Y, Yamanaka H. Interstitial lung disease with ANCA-associated vasculitis. Clin Med Insights Circ Respir Pulm Med 2015; 9(Suppl 1):51-6. doi: 10.4137/CCRPM.S23314.
doi: 10.4137/CCRPM.S23314 |
| [5] |
Jennette JC, Falk RJ, Andrassy K, et al. Nomenclature of systemic vasculitides. Proposal of an international consensus conference. Arthritis Rheum 1994; 37(2):187-92. doi: 10.1002/art.1780370206.
doi: 10.1002/art.1780370206 |
| [6] |
Suppiah R, Robson JC, Grayson PC, et al. 2022 American College of Rheumatology/European Alliance of Associations for Rheumatology classification criteria for microscopic polyangiitis. Ann Rheum Dis 2022; 81(3):321-6. doi: 10.1136/annrheumdis-2021-221796.
doi: 10.1136/annrheumdis-2021-221796 pmid: 35110332 |
| [7] |
Mukhtyar C, Lee R, Brown D, et al. Modification and validation of the Birmingham Vasculitis Activity Score (version 3). Ann Rheum Dis 2009; 68(12):1827-32. doi: 10.1136/ard.2008.101279
doi: 10.1136/ard.2008.101279 pmid: 19054820 |
| [8] |
West S, Arulkumaran N, Ind PW, et al. Diffuse alveolar haemorrhage in ANCA-associated vasculitis. Intern Med 2013; 52(1):5-13. doi: 10.2169/internalmedicine.52.8863.
doi: 10.2169/internalmedicine.52.8863 |
| [9] |
Ahn JK, Hwang JW, Lee J, et al. Clinical features and outcome of microscopic polyangiitis under a new consensus algorithm of ANCA-associated vasculitides in Korea. Rheumatol Int 2012; 32(10):2979-86. doi: 10.1007/s00296-011-2079-4.
doi: 10.1007/s00296-011-2079-4 pmid: 21898069 |
| [10] |
Furuta S, Chaudhry AN, Hamano Y, et al. Comparison of phenotype and outcome in microscopic polyangiitis between Europe and Japan. J Rheumatol 2014; 41(2):325-33. doi: 10.3899/jrheum.130602.
doi: 10.3899/jrheum.130602 pmid: 24429174 |
| [11] |
Kawai H, Banno S, Kikuchi S, et al. Retrospective analysis of factors predicting end-stage renal failure or death in patients with microscopic polyangiitis with mainly renal involvement. Clin Exp Nephrol 2014; 18(5):795-802. doi: 10.1007/s10157-013-0926-1.
doi: 10.1007/s10157-013-0926-1 pmid: 24363101 |
| [12] |
Homma S, Suzuki A, Sato K. Pulmonary involvement in ANCA-associated vasculitis from the view of the pulmonologist. Clin Exp Nephrol 2013; 17(5):667-71. doi: 10.1007/s10157-012-0710-7.
doi: 10.1007/s10157-012-0710-7 pmid: 23188194 |
| [13] |
Chen Y, Gao E, Yang L, et al. Long-term outcome of mycophenolate mofetil treatment for patients with microscopic polyangiitis: an observational study in Chinese patients. Rheumatol Int 2016; 36(7):967-74. doi: 10.1007/s00296-016-3492-5.
doi: 10.1007/s00296-016-3492-5 pmid: 27169414 |
| [14] |
Abe Y, Tamura N, Yang KS, et al. Predictive factors for mortality in elderly Japanese patients with severe microscopic polyangiitis: a retrospective single-center study. Mod Rheumatol 2017; 27(2):315-9. doi: 10.1080/14397595.2016.1205778.
doi: 10.1080/14397595.2016.1205778 pmid: 27400379 |
| [15] |
Lin Y, Zheng W, Tian X, et al. Antineutrophil cytoplasmic antibody-associated vasculitis complicated with diffuse alveolar hemorrhage: a study of 12 cases. J Clin Rheumatol 2009; 15(7):341-4. doi: 10.1097/RHU.0b013e3181b59581.
doi: 10.1097/RHU.0b013e3181b59581 pmid: 20009969 |
| [16] |
Hirayama K, Kobayashi M, Usui J, et al. Pulmonary involvements of anti-neutrophil cytoplasmic autoantibody-associated renal vasculitis in Japan. Nephrol Dial Transplant 2015; 30 (Suppl 1):i83-93. doi: 10.1093/ndt/gfu385.
doi: 10.1093/ndt/gfu385 |
| [17] |
Hogan SL, Nachman PH, Wilkman AS, et al. Prognostic markers in patients with antineutrophil cytoplasmic autoantibody-associated microscopic polyangiitis and glomerulonephritis. J Am Soc Nephrol 1996; 7(1):23-32. doi: 10.1681/ASN.V7123.
doi: 10.1681/ASN.V7123 pmid: 8808106 |
| [18] | Kostianovsky A, Hauser T, Pagnoux C, et al. Alveolar haemorrhage in ANCA-associated vasculitides: 80 patients’ features and prognostic factors. Clin Exp Rheumatol 2012; 30(1 Suppl 70):S77-82. |
| [19] |
Nada AK, Torres VE, Ryu JH, et al. Pulmonary fibrosis as an unusual clinical manifestation of a pulmonary-renal vasculitis in elderly patients. Mayo Clin Proc 1990; 65(6):847-56. doi: 10.1016/s0025-6196(12)62575-0.
doi: 10.1016/s0025-6196(12)62575-0 pmid: 2195245 |
| [20] |
Schirmer JH, Wright MN, Vonthein R, et al. Clinical presentation and long-term outcome of 144 patients with microscopic polyangiitis in a monocentric German cohort. Rheumatology (Oxford) 2016; 55(1):71-9. doi: 10.1093/rheumatology/kev286.
doi: 10.1093/rheumatology/kev286 pmid: 26297628 |
| [21] |
Arulkumaran N, Periselneris N, Gaskin G, et al. Interstitial lung disease and ANCA-associated vasculitis: a retrospective observational cohort study. Rheumatology (Oxford) 2011; 50(11):2035-43. doi: 10.1093/rheumatology/ker236.
doi: 10.1093/rheumatology/ker236 pmid: 21873269 |
| [22] |
Li ZY, Chang DY, Zhao MH, et al. Predictors of treatment resistance and relapse in antineutrophil cytoplasmic antibody-associated vasculitis: a study of 439 cases in a single Chinese center. Arthritis Rheumatol 2014; 66(7):1920-6. doi: 10.1002/art.38621.
doi: 10.1002/art.38621 |
| [23] |
Kadura S, Raghu G. Antineutrophil cytoplasmic antibody-associated interstitial lung disease: a review. Eur Respir Rev 2021; 30(162): 210123. doi: 10.1183/16000617.0123-2021.
doi: 10.1183/16000617.0123-2021 |
| [24] | Sebastiani M, Manfredi A, Vacchi C, et al. Epidemiology and management of interstitial lung disease in ANCA-associated vasculitis. Clin Exp Rheumatol 2020; 38 (2):221-31. |
| [25] |
Hervier B, Pagnoux C, Agard C, et al. Pulmonary fibrosis associated with ANCA-positive vasculitides. Retrospective study of 12 cases and review of the literature. Ann Rheum Dis 2009; 68(3):404-7. doi: 10.1136/ard.2008.096131.
doi: 10.1136/ard.2008.096131 pmid: 18957485 |
| [26] |
Tzelepis GE, Kokosi M, Tzioufas A, et al. Prevalence and outcome of pulmonary fibrosis in microscopic polyangiitis. Eur Respir J 2010; 36(1):116-21. doi: 10.1183/09031936.00110109.
doi: 10.1183/09031936.00110109 pmid: 19926741 |
| [27] |
Comarmond C, Crestani B, Tazi A, et al. Pulmonary fibrosis in antineutrophil cytoplasmic antibodies (ANCA)-associated vasculitis: a series of 49 patients and review of the literature. Medicine (Baltimore) 2014; 93(24):340-9. doi: 10.1097/MD.0000000000000217.
doi: 10.1097/MD.0000000000000217 |
| [28] |
Fernandez Casares M, Gonzalez A, Fielli M, et al. Microscopic polyangiitis associated with pulmonary fibrosis. Clin Rheumatol 2015; 34(7):1273-7. doi: 10.1007/s10067-014-2676-1.
doi: 10.1007/s10067-014-2676-1 pmid: 24863847 |
| [29] |
Huang H, Wang YX, Jiang CG, et al. A retrospective study of microscopic polyangiitis patients presenting with pulmonary fibrosis in China. BMC Pulm Med 2014; 14:8. doi: 10.1186/1471-2466-14-8.
doi: 10.1186/1471-2466-14-8 pmid: 24468083 |
| [30] |
Hozumi H, Kono M, Hasegawa H, et al. Clinical significance of interstitial lung disease and its acute exacerbation in microscopic polyangiitis. Chest 2021; 159(6):2334-45. doi: 10.1016/j.chest.2021.01.083.
doi: 10.1016/j.chest.2021.01.083 pmid: 33571506 |
| [31] |
Kagiyama N, Takayanagi N, Kanauchi T, et al. Antineutrophil cytoplasmic antibody-positive conversion and microscopic polyangiitis development in patients with idiopathic pulmonary fibrosis. BMJ Open Respir Res 2015; 2(1):e000058. doi: 10.1136/bmjresp-2014-000058.
doi: 10.1136/bmjresp-2014-000058 |
| [32] |
Zhou P, Li Z, Gao L, et al. Pulmonary involvement of ANCA-associated vasculitis in adult Chinese patients. BMC Pulm Med 2022; 22(1): 35. doi: 10.1186/s12890-022-01829-y.
doi: 10.1186/s12890-022-01829-y pmid: 35022009 |
| [33] |
Sarica SH, Dhaun N, Sznajd J, et al. Characterizing infection in anti-neutrophil cytoplasmic antibody-associated vasculitis: results from a longitudinal, matched-cohort data linkage study. Rheumatology (Oxford) 2020; 59(10):3014-22. doi: 10.1093/rheumatology/keaa070.
doi: 10.1093/rheumatology/keaa070 pmid: 32159801 |
| [34] |
Lauque D, Cadranel J, Lazor R, et al. Microscopic polyangiitis with alveolar hemorrhage. A study of 29 cases and review of the literature. Medicine (Baltimore) 2000; 79(4):222-33. doi: 10.1097/00005792-200007000-00003.
doi: 10.1097/00005792-200007000-00003 |
| [35] |
Matsuda S, Kotani T, Suzuka T, et al. Evaluation of poor prognostic factors of respiratory related death in microscopic polyangiitis complicated by interstitial lung disease. Sci Rep 2021; 11(1):1490. doi: 10.1038/s41598-021-81311-7.
doi: 10.1038/s41598-021-81311-7 pmid: 33452394 |
| [36] |
Kida T, Tanaka T, Yokota I, et al. Association between preexisting lung involvements and the risk of diffuse alveolar hemorrhage in patients with microscopic polyangiitis: a multi-center retrospective cohort study. Mod Rheumatol 2020; 30(2):338-44. doi: 10.1080/14397595.2019.1601855.
doi: 10.1080/14397595.2019.1601855 pmid: 30929541 |
| [1] | 张雪, 吴林格尔, 黄会真, 张越伦, 鲁志龙, 田亚杰, 申乐, 黄宇光. 麻醉相关气道不良事件相关因素分析:一项2009至2022年单中心回顾性病例对照研究[J]. Chinese Medical Sciences Journal, 2022, 37(4): 287-292. |
| [2] | 马璐璐, 虞雪融, 翁习生, 林进, 金今, 钱文伟, 黄宇光. 全膝关节置换术患者术后严重并发症的危险因素[J]. Chinese Medical Sciences Journal, 2022, 37(4): 303-308. |
| [3] | 林立金, 柳叶茂, 秦娟娟, 雷昉, 汪文鑫, 黄学伟, 刘伟芳, 张兴元, 折志刚, 张鹏, 张晓晶, 金朝霞, 李红良. 1990-2030年全球和区域感染性心内膜炎的疾病负担及其归因危险因素的趋势和预测[J]. Chinese Medical Sciences Journal, 2022, 37(3): 181-194. |
| [4] | 吴斌,周江华,汪文鑫,杨慧琳,夏盟,张丙宏,折志刚,李红良. 高脂血症与新型冠状病毒肺炎住院患者28天全因死亡率的关系[J]. Chinese Medical Sciences Journal, 2021, 36(1): 17-26. |
| [5] | 尹俊雄, 余诚, 魏丽霞, 余传勇, 刘红星, 杜明洋, 孙丰, 王崇骏, 王小姗. 基于机器学习的脑卒中高危人群无症状性颈动脉狭窄的检测研究[J]. Chinese Medical Sciences Journal, 2020, 35(4): 297-305. |
| [6] | 李涛, 杨立, 张卫国, 罗春才, 黄自立, 李金锋, 李欣. 6 4层多排CT对冠状动脉旁路移植术后的中期随访:影响桥血管通畅性的危险因素研究[J]. Chinese Medical Sciences Journal, 2018, 33(2): 69-76. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
|