Chinese Medical Sciences Journal ›› 2020, Vol. 35 ›› Issue (4): 357-365.doi: 10.24920/003659
• Review • Previous Articles Next Articles
Wnt5a Plays Controversial Roles in Cancer Progression
Xu Chen1, Hongling Liu2, Fenghui Zhao1, *( ), Zongxian Jiao3, Jinsui Wang1, Yamei Dang1
), Zongxian Jiao3, Jinsui Wang1, Yamei Dang1
- 1Department of Pathology, Gansu Provincial Hospital, Lanzhou 730000, China
2Department of Occupational Medicine, the Third Gansu Provincial Hospital, Lanzhou 730000, China
3Pathological Institution of Basic Medical College, Lanzhou University, Lanzhou 730000, China
-
Received:2019-09-12Accepted:2020-02-04Published:2020-12-31Online:2020-10-28 -
Contact:Fenghui Zhao E-mail:zhaofh0931@163.com
Cite this article
Xu Chen, Hongling Liu, Fenghui Zhao, Zongxian Jiao, Jinsui Wang, Yamei Dang. Wnt5a Plays Controversial Roles in Cancer Progression[J].Chinese Medical Sciences Journal, 2020, 35(4): 357-365.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

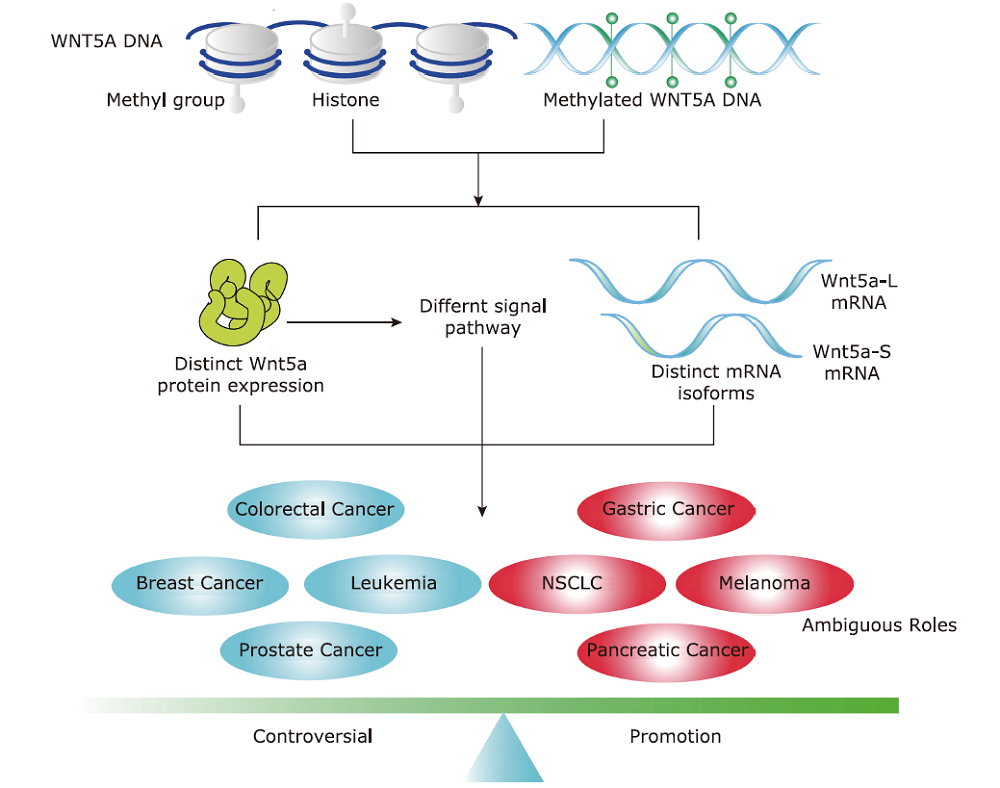
Figure 1.
Wnt signaling pathway. The two groups of Wnt factors regulate the β-catenin, calcium and planar cell polarity (PCP)/convergent extension (CE) pathways. APC, adenomatous polyposis coli; CAMK Ⅱ, calmodulin dependent protein kinase Ⅱ; LRP, lipoprotein receptor-related protein co-receptor; DAG, diacylglycerol; Dvl, dishevelled; IP3, inositol 1,4,5-trisphosphate; JNK, Jun N-terminal kinase; NFAT, nuclear factor of activated T cell; PIP2, phosphatidylinositol 4,5-bisphosphate; PLC, proteins and phospholipase C; PKC, protein kinase C; Ror2, receptor tyrosine kinase-like orphan receptor 2; TCF, T-cell factor; LEF, lymphoid enhancer factor."
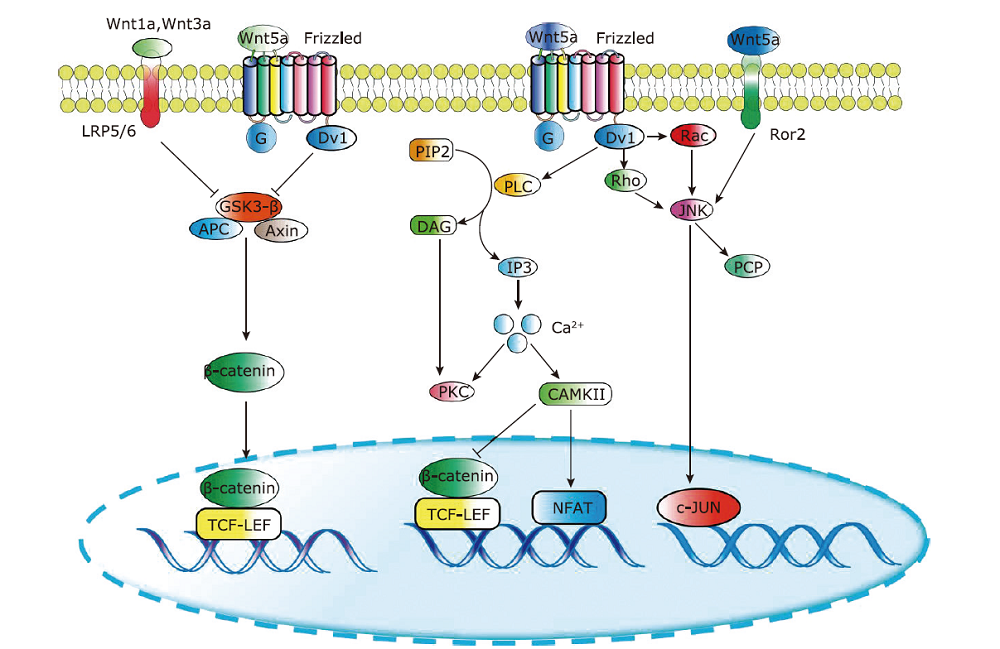
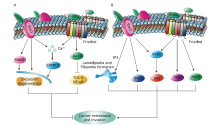
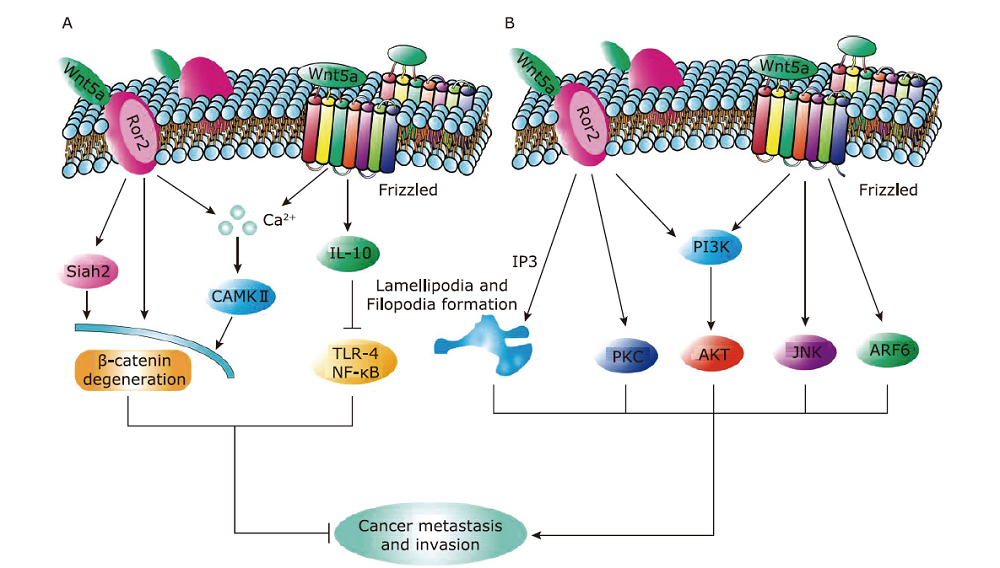
Figure 2.
Ambiguous roles of Wnt5a in cancer metastasis. (A) Inhibition of metastasis in cancer. (B) Promotion of metastasis in cancer. ARF6, ADP-ribosylation factor 6; NF-kB, nuclear factor-kB; IL-10, interleukin 10; PI3K, phosphatidylinositol 3‐kinase; Siah2, absentia homologues 2; TLR-4, toll-like receptor-4."
| 1. |
Wang X, Zhao X, Yi Z, et al. WNT5A promotes migration and invasion of human osteosarcoma cells via SRC/ERK/MMP-14 pathway. Cell Bio Int 2018; 42(5):598-607. doi: 10.1002/cbin.10936.
doi: 10.1002/cbin.v42.5 |
| 2. |
Wang W, Yu X, Wu C, et al. Differential effects of Wnt5a on the proliferation, differentiation and inflammatory response of keratinocytes. Mol Med Rep 2017; 17(3):4043-48. doi: 10.3892/mmr.2017.8358.
doi: 10.3892/mmr.2017.8358 pmid: 29286164 |
| 3. |
Feng C, Liang S, Harold V. Wnt signals across the plasma membrane to activate the beta-catenin pathway by forming oligomers containing its receptors, Frizzled and LRP. Development 2004; 131:5103-15. doi: 10.1089/scd.2015.0367.
doi: 10.1242/dev.01318 pmid: 15459103 |
| 4. |
Zins K, Schäfer R, Paulus P, et al. Frizzled2 signaling regulates growth of high-risk neuroblastomas by interfering with β-catenin-dependent and β-catenin-independent signaling pathways. Oncotarget 2016; 7(29):46187-202. doi: 10.18632/oncotarget.10070.
pmid: 27323822 |
| 5. |
Keller KC, Ding H, Tieu R, et al. Wnt5a Supports Osteogenic Lineage Decisions in Embryonic Stem Cells. Stem Cells Dev 2016; 25(13):1020-32. doi: 10.1089/scd.2015.0367.
pmid: 26956615 |
| 6. |
Abedini A, Zamberlam G, Lapointe E, et al. WNT5a is required for normal ovarian follicle development and antagonizes gonadotropin responsiveness in granulosa cells by suppressing canonical WNT signaling. Faseb J 2016; 30(4):1534-47. doi: 10.1096/fj.15-280313.
doi: 10.1096/fj.15-280313 pmid: 26667040 |
| 7. |
Saling M, Duckett JK, Ackers I, et al. Wnt5a/planar cell polarity signaling pathway in urothelial carcinoma, a potential prognostic biomarker. Oncotarget 2017; 8(19):31655-65. doi: 10.18632/oncotarget.15877.
doi: 10.18632/oncotarget.15877 pmid: 28427201 |
| 8. |
Zhong Z, Shan M, Wang J, et al. Decreased Wnt5a Expression is a Poor Prognostic Factor in Triple-Negative Breast Cancer. Med Sci Monit 2016; 22:1-7. doi: 10.12659/MSM.894821
pmid: 26721633 |
| 9. |
Matthieu B, Jean B, Terry G, et al. WNT5A encodes two isoforms with distinct functions in cancers. Plos One 2013; 8:e80526. doi: 10.1371/journal.pone.0080526.
doi: 10.1371/journal.pone.0080526 pmid: 24260410 |
| 10. |
Gavin BJ, Mcmahon JA, Mcmahon AP. Expression of multiple novel Wnt-1/int-1-related genes during fetal and adult mouse development. Genes Dev 1990; 4(12B):2319-32. doi: 10.1101/gad.4.12b.2319.
pmid: 2279700 |
| 11. | Parr BA, Mcmahon AP. Wnt genes and vertebrate development. Cur Opinion Genet Develop 1994; 4(4):523-8. doi: 10.1016/0959-437X(94)90067-D. |
| 12. |
Clark CC, Cohen I, Eichstetter I, et al. Molecular cloning of the human proto-oncogene Wnt-5A and mapping of the gene (WNT5A) to chromosome 3p14-p21. Genomics 1993; 18(2):249-60. doi: 10.1006/geno.1993.1463.
doi: 10.1006/geno.1993.1463 pmid: 8288227 |
| 13. |
Lin HK, Lin HH, Chiou YW, et al. Caveolin-1 down-regulation is required for Wnt5a-Frizzled 2 signalling in Ha-RasV12-induced cell transformation. J Cellular Molecular Med 2018; 22(5):2631-43. doi: 10.1111/jcmm.13531.
doi: 10.1111/jcmm.2018.22.issue-5 |
| 14. |
Wang HY, Liu T, Malbon CC. Structure-function analysis of Frizzleds. Cell Signal 2006; 18(7):934-41. doi: 10.1016/j.cellsig.2005.12.008.
doi: 10.1016/j.cellsig.2005.12.008 |
| 15. |
Bischoff DS, Zhu JH, Makhijani NS, et al. Induction of CXC chemokines in human mesenchymal stem cells by stimulation with secreted frizzled-related proteins through non-canonical Wnt signaling. World J Stem Cells 2015; 7(11):1262-73. doi: 10.4252/wjsc.v7.i11.1262.
doi: 10.4252/wjsc.v7.i11.1262 pmid: 26730270 |
| 16. | Uehara S, Udagawa N, Mukai H, et al. Protein kinase N3 promotes bone resorption by osteoclasts in response to Wnt5a-Ror2 signaling. Sci Signaling 2017; 10(494):e0023. doi: 10.1126/scisignal.aan0023. |
| 17. |
Skaria T, Bachli E, Schoedon G. Wnt5A/Ryk signaling critically affects barrier function in human vascular endothelial cells. Cell Adh Migr 2016; 11(1):24-38. doi: 10.1080/19336918.2016.1178449.
pmid: 27159116 |
| 18. |
Adamo A, Fiore D, Martino FD, et al. RYK promotes the stemness of glioblastoma cells via the WNT/ β-catenin pathway. Oncotarget 2017; 8(26):13476-87. doi: 10.18632/oncotarget.14564.
doi: 10.18632/oncotarget.v8i8 |
| 19. |
Sakisaka Y, Kanaya S, Nakamura T, et al. p38 MAP kinase is required for Wnt3a-mediated osterix expression independently of Wnt-LRP5/6-GSK3β signaling axis in dental follicle cells. Biochem Biophys Res Commun 2016; 478(2):527-32. doi: 10.1016/j.bbrc.2016.07.076.
doi: 10.1016/j.bbrc.2016.07.076 pmid: 27450807 |
| 20. |
Lai K, Kweon SM, Chi F, et al. Abstract 4331: Novel Wnt-SCD-LRP5/6 pathway linking liver fibrosis to cancer. Cancer Res 2017; 77. doi: 10.1158/1538-7445.AM2017-4331.
doi: 10.1158/0008-5472.CAN-17-2066 pmid: 29097609 |
| 21. |
Kim JG, Kim MJ, Choi WJ, et al. Wnt3A Induces GSK-3β Phosphorylation and β-Catenin Accumulation Through RhoA/ROCK. J cell physiol 2017; 232(5). 1104-13. doi: 10.1002/jcp.25572.
doi: 10.1002/jcp.25572 pmid: 27575935 |
| 22. |
Daniels DL, Weis WI. β-catenin directly displaces Groucho/TLE repressors from Tcf/Lef in Wnt-mediated transcription activation. Nat Struct Mol Biol 2005; 12(4):364-71. doi: 10.1038/nsmb912.
pmid: 15768032 |
| 23. |
Yu L, Liu S, Zhao Z, et al. Extracorporeal shock wave rebuilt subchondral bone in vivo and activated Wnt5a/Ca2+ signaling in vitro. Biomed Res Int 2017; 2017:1404650. doi: 10.1155/2017/1404650.
doi: 10.1155/2017/1404650 pmid: 29164146 |
| 24. | Hatziapostolou M, Koukos G, Polytarchou C, et al. Tumor progression locus 2 mediates signal-induced increases in cytoplasmic calcium and cell migration. Sci Signal 2011; 4(187):e55. doi: 10.1126/scisignal.2002006. |
| 25. |
Ho HYH, Susman MW, Bikoff JB, et al. Wnt5a-Ror-Dishevelled signaling constitutes a core developmental pathway that controls tissue morphogenesis. Proc Natl Acad Sci U S A 2012; 109(1):4044-51. doi: 10.1073/pnas.1200421109.
doi: 10.1073/pnas.1200421109 |
| 26. |
Martineau X, Abed É, Martel-Pelletier J, et al. Alteration of Wnt5a expression and of the non-canonical Wnt/PCP and Wnt/PKC-Ca2+ pathways in human osteoarthritis osteoblasts. Plos One 2017; 12(8):e0180711. doi: 10.1371/journal.pone.0180711.
doi: 10.1371/journal.pone.0180711 pmid: 28777797 |
| 27. |
Chen M, Zhong W, Hu Y, et al. Wnt5a/FZD5/CaMKII signaling pathway mediates the effect of BML-111 on inflammatory reactions in sepsis. Int J Clin Exp Med 2015; 8(10):17824-9.
pmid: 26770375 |
| 28. |
Chattopadhyay S, Chatterjee R, Law S. Noncanonical Wnt5a-Ca2+-NFAT signaling axis in pesticide induced bone marrow aplasia mouse model: A study to explore the novel mechanism of pesticide toxicity. Environ Toxicol 2016(10); 31:1163-75. doi: 10.1002/tox.22123.
doi: 10.1002/tox.22123 pmid: 25846497 |
| 29. |
Saling M, Duckett JK, Ackers I, et al. Wnt5a/planar cell polarity signaling pathway in urothelial carcinoma, a potential prognostic biomarker. Oncotarget 2017; 8(19):31655-65. doi: 10.18632/oncotarget.15877.
doi: 10.18632/oncotarget.15877 pmid: 28427201 |
| 30. |
Hasegawa D, Wada N, Yoshida S, et al. Wnt5a suppresses osteoblastic differentiation of human periodontal ligament stem cell-like cells via Ror2/JNK signaling. J cell physiol 2018; 233(2):1752-62. doi: 10.1002/jcp.26086.
doi: 10.1002/jcp.26086 pmid: 28681925 |
| 31. |
Yu J, Chen Y, Chen L, et al. Cirmtuzumab inhibits ibrutinib-resistant, Wnt5a-induced Rac1 activation and proliferation in mantle cell lymphoma. Oncotarget 2018; 9(37):24731-6. doi: 10.18632/oncotarget.25340.
doi: 10.18632/oncotarget.25340 pmid: 29872501 |
| 32. |
Yu J, Chen L, Chen Y, et al. Wnt5a induces ROR1 to associate with 14-3-3ζ for enhanced chemotaxis and proliferation of chronic lymphocytic leukemia cells. Leukemia 2017; 31(12):2608-14. doi: 10.1038/leu.2017.132.
doi: 10.1038/leu.2017.132 pmid: 28465528 |
| 33. |
Prasad CP, Södergren K, Andersson T. Reduced production and uptake of lactate are essential for the ability of WNT5A signaling to inhibit breast cancer cell migration and invasion. Oncotarget 2017; 8(42):71471-88. doi: 10.18632/oncotarget.17277.
pmid: 29069720 |
| 34. |
Li J, Ying J, Fan Y, et al. WNT5A antagonizes WNT/β-catenin signaling and is frequently silenced by promoter CpG methylation in esophageal squamous cell carcinoma. Cancer Biol Ther 2010; 10(6):617-24. doi: 10.4161/cbt.10.6.12609.
doi: 10.4161/cbt.10.6.12609 pmid: 20603606 |
| 35. |
Ann EJ, Kim HY, Seo MS, et al. Wnt5a controls Notch1 signaling through CaMKII-mediated degradation of the SMRT corepressor protein. J Biol Chem 2012; 287(44):36814-29. doi: 10.1074/jbc.M112.356048.
doi: 10.1074/jbc.M112.356048 pmid: 22888005 |
| 36. |
R John M, Madeline H. Wnt5a secretion stimulated by the extracellular calcium-sensing receptor inhibits defective Wnt signaling in colon cancer cells. Am J Physiol Gastrointest Liver Physiol 2007; 293(1):G403-11. doi: 10.1152/ajpgi.00119.2007.
doi: 10.1152/ajpgi.00119.2007 pmid: 17463182 |
| 37. |
Voloshanenko O, Schwartz U, Kranz D, et al. β-catenin-independent regulation of Wnt target genes by RoR2 and ATF2/ATF4 in colon cancer cells. Sci Rep 2018; 8(1):3178. doi: 10.1038/s41598-018-20641-5.
pmid: 29453334 |
| 38. |
Bergenfelz C, Medrek C, Ekström E, et al. Wnt5a induces a tolerogenic phenotype of macrophages in sepsis and breast cancer patients. J Immunol 2012; 188(11):5448-58. doi: 10.4049/jimmunol.1103378.
doi: 10.4049/jimmunol.1103378 pmid: 22547701 |
| 39. |
Zhang W, Yan Y, Gu M, et al. High expression levels of Wnt5a and Ror2 in laryngeal squamous cell carcinoma are associated with poor prognosis. Oncol letters 2017; 14(2):2232-8. doi: 10.3892/ol.2017.6386.
doi: 10.3892/ol.2017.6386 |
| 40. |
Dai B, Yan T, Zhang A. ROR2 receptor promotes the migration of osteosarcoma cells in response to Wnt5a. Cancer Cell Int 2017; 17:112. doi: 10.1186/s12935-017-0482-y.
doi: 10.1186/s12935-017-0482-y pmid: 29213214 |
| 41. |
Oishi I, Suzuki H, Onishi N, et al. The receptor tyrosine kinase Ror2 is involved in non-canonical Wnt5a/JNK signaling pathway. Genes Cells 2003; 8(7):645-54. doi: 10.1046/j.1365-2443.2003.00662.x.
doi: 10.1046/j.1365-2443.2003.00662.x pmid: 12839624 |
| 42. |
Nam S, Chung JW, Yang JY. WNT5A correlates with clinicopathological characteristics in gastric cancer: a meta-analysis. Cell Physiol Biochem 2017; 41(1):33-40. doi: 10.1159/000455934.
doi: 10.1159/000455934 pmid: 28135710 |
| 43. |
Ara H, Takagishi M, Enomoto A, et al. Role for Daple in non-canonical Wnt signaling during gastric cancer invasion and metastasis. Cancer Sci 2016; 107(2):133-9. doi: 10.1111/cas.12848.
doi: 10.1111/cas.12848 pmid: 26577606 |
| 44. |
Liu J, Zhang Y, Xu R, et al. PI3K/Akt-dependent phosphorylation of GSK3β and activation of RhoA regulate Wnt5a-induced gastric cancer cell migration. Cell Signal 2013; 25(2):447-56. doi: 10.1016/j.cellsig.2012.10.012.
doi: 10.1016/j.cellsig.2012.10.012 pmid: 23123500 |
| 45. |
Hanaki H, Yamamoto H, Sakane H, et al. An anti-Wnt5a antibody suppresses metastasis of gastric cancer cells in vivo by inhibiting receptor-mediated endocytosis. Mol Cancer Ther 2012; 11(2):298-307. doi: 10.1158/1535-7163.mct-11-0682.
doi: 10.1158/1535-7163.MCT-11-0682 pmid: 22101459 |
| 46. |
Yamamoto H, Kitadai Y, Naohide Oue, et al. Laminin γ2 Mediates Wnt5a-Induced Invasion of Gastric Cancer Cells. Gastroenterology 2009; 137(1):242-52. doi: 10.1053/j.gastro.2009.02.003.
doi: 10.1053/j.gastro.2009.02.003 |
| 47. |
Kanzawa M, Semba S, Hara S, et al. WNT5A is a key regulator of the epithelial-mesenchymal transition and cancer stem cell properties in human gastric carcinoma cells. Pathobiology 2013; 80(5):235-44. doi: 10.1159/000346843.
doi: 10.1159/000346843 pmid: 23615002 |
| 48. |
Zhang Y, Du J, Zheng J, et al. EGF-reduced Wnt5a transcription induces epithelial-mesenchymal transition via Arf6-ERK signaling in gastric cancer cells. Oncotarget 2015; 6:7244-61. doi: 10.18632/oncotarget.3133.
doi: 10.18632/oncotarget.3133 pmid: 25779663 |
| 49. |
Takiguchi G, Nishita M, Kurita K, et al. Wnt5a-Ror2 signaling in mesenchymal stem cells promotes proliferation of gastric cancer cells by activating CXCL16-CXCR6 axis. Cancer Sci 2016; 107(9):290-7. doi: 10.1111/cas.12871.
doi: 10.1111/cas.2016.107.issue-3 |
| 50. |
Dejmek J, Dejmek A, Säfholm A, et al. Wnt-5a protein expression in primary Dukes B colon cancers identifies a subgroup of patients with good prognosis. Cancer Res 2005; 65:9142-6. doi: 10.1158/0008-5472.CAN-05-1710.
doi: 10.1158/0008-5472.CAN-05-1710 pmid: 16230369 |
| 51. |
Tao J, Shi L, Huang L, et al. EZH2 is involved in silencing of WNT5A during epithelial-mesenchymal transition of colon cancer cell line. J Cancer Res Clin Oncol 2017; 143(11):2211-9. doi: 10.1007/s00432-017-2479-2.
doi: 10.1007/s00432-017-2479-2 pmid: 28748258 |
| 52. |
Mehdawi LM, Prasad CP, Ehrnström R, et al. Non-canonical WNT5A signaling up-regulates the expression of the tumor suppressor 15-PGDH and induces differentiation of colon cancer cells. Mol Oncol 2016; 10:1415-29. doi: 10.1016/j.molonc.2016.07.011.
doi: 10.1016/j.molonc.2016.07.011 pmid: 27522468 |
| 53. |
Cheng R, Sun B, Liu Z, et al. Wnt5a suppresses colon cancer by inhibiting cell proliferation and epithelial-mesenchymal transition. J Cell Physiol 2014; 229(12):1908-17. doi: 10.1002/jcp.24566.
doi: 10.1002/jcp.24566 pmid: 24464650 |
| 54. |
Wang J, Wang X, Liu F, et al. microRNA-335 inhibits colorectal cancer HCT116 cells growth and epithelial-mesenchymal transition (EMT) process by targeting Twist1. Pharmazie 2017; 72(8):475-81. doi: 10.1691/ph.2017.7489.
doi: 10.1691/ph.2017.7489 pmid: 29441907 |
| 55. |
Bakker ERM, Das AM, Helvensteijn W, et al. Wnt5a promotes human colon cancer cell migration and invasion but does not augment intestinal tumorigenesis in Apc 1638N mice. Carcinogenesis 2013; 34(11):2629-38. doi: 10.1093/carcin/bgt215.
doi: 10.1093/carcin/bgt215 pmid: 23764752 |
| 56. |
Huang TC, Lee PT, Wu MH, et al. Distinct roles and differential expression levels of Wnt5a mRNA isoforms in colorectal cancer cells. PloS One 2017; 12(8):e0181034. doi: 10.1371/journal.pone.0181034.
doi: 10.1371/journal.pone.0181034 pmid: 28859077 |
| 57. |
Antonia SJ, Villegas A, Daniel D, et al. Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med 2017; 377(20):1919-29. doi: 10.1056 /nejmoa1709937.
doi: 10.1056/NEJMoa1709937 pmid: 28885881 |
| 58. |
Liu M, Zhang Y, Zhang J, et al. MicroRNA-1253 suppresses cell proliferation and invasion of non-small-cell lung carcinoma by targeting WNT5A. Cell Death Dis 2018; 9(2):189. doi: 10.1038/s41419-017-0218-x.
doi: 10.1038/s41419-017-0218-x pmid: 29415994 |
| 59. |
Wang B, Tang Z, Gong H, et al. Wnt5a promotes epithelial-to-mesenchymal transition and metastasis in non-small-cell lung cancer. Biosci Rep 2017; 37(6): BSR20171092. doi: 10.1042/BSR20171092.
doi: 10.1042/BSR20171092 pmid: 29054966 |
| 60. | Huang Cl, Liu D, Nakano J, et al. Wnt5a expression is associated with the tumor proliferation and the stromal vascular endothelial growth factor: An expression in non-small-cell lung cancer. J Clinc Oncol 2005; 23(34):8765-73. doi: 10.1200/jco.2005.02.2871. |
| 61. |
Whang YM, Jo U, Sung JS, et al. Wnt5a is associated with cigarette smoke-related lung carcinogenesis via protein kinase C. PLoS One 2013; 8(1):e53012. doi: 10.1371/journal.pone.0053012.
doi: 10.1371/journal.pone.0053012 pmid: 23349696 |
| 62. |
Yang J, Zhang K, Wu J, et al. Wnt5a increases properties of lung cancer stem cells and resistance to cisplatin through activation of Wnt5a/PKC signaling pathway. Stem Cells Int 2016; 2016:1690896. doi: 10.1155/2016/1690896.
doi: 10.1155/2016/1690896 pmid: 27895670 |
| 63. |
Kobayashi Y, Kadoya T, Amioka A, et al. Wnt5a-induced cell migration is associated with the aggressiveness of estrogen receptor-positive breast cancer. Oncotarget 2018; 9(30):20979-92. doi: 10.18632/oncotarget.24761.
doi: 10.18632/oncotarget.24761 pmid: 29765514 |
| 64. |
Prasad CP, Chaurasiya SK, Guilmain W, et al. WNT5A signaling impairs breast cancer cell migration and invasion via mechanisms independent of the epithelial-mesenchymal transition. J Exp Clin Cancer Res 2016; 35(1):144. doi: 10.1186/s13046-016-0421-0.
doi: 10.1186/s13046-016-0421-0 pmid: 27623766 |
| 65. |
Prasad CP, Chaurasiya SK, Axelsson L, et al. WNT-5A triggers Cdc42 activation leading to an ERK1/2 dependent decrease in MMP9 activity and invasive migration of breast cancer cells. Mol Oncol 2013; 7(5):870-83. doi: 10.1016/j.molonc.2013.04.005.
doi: 10.1016/j.molonc.2013.04.005 pmid: 23727359 |
| 66. |
Chrysanthou E, Gorringe KL, Joseph C, et al. Phenotypic characterisation of breast cancer: the role of CDC42. Breast Cancer Res Treat 2017; 164(2):317-25. doi: 10.1007/s10549-017-4267-8.
doi: 10.1007/s10549-017-4267-8 pmid: 28451966 |
| 67. |
Jiang W, Crossman DK, Mitchell EH, et al. WNT5A inhibits metastasis and alters splicing of Cd44 in breast cancer cells. PLoS One 2013; 8(3):e58329. doi: 10.1371/journal.pone.0058329.
doi: 10.1371/journal.pone.0058329 pmid: 23484019 |
| 68. |
Han B, Zhou B, Qu Y, et al. FOXC1-induced non-canonical WNT5A-MMP7 signaling regulates invasiveness in triple-negative breast cancer. Oncogene 2018; 37(10):1399-408. doi: 10.1038/s41388-017-0021-2.
doi: 10.1038/s41388-017-0021-2 pmid: 29249801 |
| 69. |
Zhu Y, Tian Y, Du J, et al. Dvl2-dependent activation of Daam1 and RhoA regulates Wnt5a-induced breast cancer cell migration. PLoS One 2012; 7(5):e37823. doi: 10.1371/journal.pone.0037823.
doi: 10.1371/journal.pone.0037823 pmid: 22655072 |
| 70. |
Zoico E, Darra E, Rizzatti V, et al. Adipocytes WNT5a mediated dedifferentiation: a possible target in pancreatic cancer microenvironment. Oncotarget 2016; 7(15):20223-35. doi: 10.18632/oncotarget.7936.
doi: 10.18632/oncotarget.7936 pmid: 26958939 |
| 71. |
Bo H, Gao L, Chen Y, et al. Upregulation of the expression of Wnt5a promotes the proliferation of pancreatic cancer cells in vitro and in a nude mouse model. Mol Med Rep 2016; 13(2):1163-71. doi: 10.3892/mmr.2015.4642.
doi: 10.3892/mmr.2015.4642 pmid: 26648282 |
| 72. |
Bo H, Zhang S, Gao L, et al. Upregulation of Wnt5a promotes epithelial-to-mesenchymal transition and metastasis of pancreatic cancer cells. BMC Cancer 2013; 13:496. doi: 10.1186/1471-2407-13-496.
doi: 10.1186/1471-2407-13-496 pmid: 24156409 |
| 73. |
Wei W, Li H, Li N, et al. WNT5A/JNK signaling regulates pancreatic cancer cells migration by phosphorylating paxillin. Pancreatology 2013; 13(4):384-92. doi: 10.1016/j.pan.2013.05.008.
doi: 10.1016/j.pan.2013.05.008 pmid: 23890137 |
| 74. |
Wei W, Sun HH, Li N, et al. WNT5A modulates cell cycle progression and contributes to the chemoresistance in pancreatic cancer cells. Hepatobiliary Pancreat Dis Int 2014; 13(5):529-38. doi: 10.1016/S1499-3872(14)60277-0.
doi: 10.1016/s1499-3872(14)60277-0 pmid: 25308364 |
| 75. |
Yamamoto H, Oue N, Sato A, et al. Wnt5a signaling is involved in the aggressiveness of prostate cancer and expression of metalloproteinase. Oncogene 2010; 29(14):2036-46. doi: 10.1038/onc.2009.496.
doi: 10.1038/onc.2009.496 pmid: 20101234 |
| 76. |
Thiele S, Zimmer A, Göbel A, et al. Role of WNT5A receptors FZD5 and RYK in prostate cancer cells. Oncotarget 2018; 9(43):27293-304. doi: 10.18632/oncotarget.25551.
doi: 10.18632/oncotarget.25551 pmid: 29930766 |
| 77. |
Lee GT, Kang DI, Ha YS, et al. Prostate cancer bone metastases acquire resistance to androgen deprivation via WNT5A-mediated BMP-6 induction. Br J Cancer 2014; 110:1634-44. doi: 10.1038/bjc.2014.23.
doi: 10.1038/bjc.2014.23 pmid: 24518599 |
| 78. |
Lee GT, Kwon SJ, Kim J, et al. WNT5A induces castration-resistant prostate cancer via CCL2 and tumour-infiltrating macrophages. Br J Cancer 2018; 118(5):670-8. doi: 10.1038/bjc.2017.451.
doi: 10.1038/bjc.2017.451 pmid: 29381686 |
| 79. |
Jin F, Qu X, Fan Q, et al. Regulation of prostate cancer cell migration toward bone marrow stromal cell-conditioned medium by Wnt5a signaling. Mol Med Rep 2013; 8(5):1486-92. doi: 10.3892/mmr.2013.1698.
doi: 10.3892/mmr.2013.1698 pmid: 24064566 |
| 80. |
Thiele S, Göbel A, Rachner TD, et al. WNT5A has anti-prostate cancer effects in vitro and reduces tumor growth in the skeleton in vivo. J Bone Miner Res 2015; 30(3):471-80. doi: 10.1002/jbmr.2362.
doi: 10.1002/jbmr.2362 pmid: 25224731 |
| 81. |
Canesin G, Evans-Axelsson S, Hellsten R, et al. Treatment with the WNT5A-mimicking peptide Foxy-5 effectively reduces the metastatic spread of WNT5A-low prostate cancer cells in an orthotopic mouse model. PLoS One 2017; 12(9):e0184418. doi: 10.1371/journal.pone.0184418.
doi: 10.1371/journal.pone.0184418 pmid: 28886116 |
| 82. | Ndoye A, Budina A, Webster M, et al. Role of autophagy in Wnt5A-mediated melanoma invasion and metastasis. Cancer Res 2015; 75(15):2906. doi: 10.1158/1538-7445.AM2015-2906. |
| 83. |
Weeraratna AT, Jiang Y, Hostetter G, et al. Wnt5a signaling directly affects cell motility and invasion of metastatic melanoma. Cancer Cell 2002; 1(3):279-88. doi: 10.1016/S1535-6108(02)00045-4.
doi: 10.1016/s1535-6108(02)00045-4 pmid: 12086864 |
| 84. |
Ekström EJ, Bergenfelz C, von Bülow V, et al. WNT5A induces release of exosomes containing pro-angiogenic and immunosuppressive factors from malignant melanoma cells. Mol Cancer 2014; 13:88. doi: 10.1186/1476-4598-13-88.
doi: 10.1186/1476-4598-13-88 pmid: 24766647 |
| 85. |
Anastas JN, Kulikauskas RM, Tamir T, et al. WNT5A enhances resistance of melanoma cells to targeted BRAF inhibitors. J Clin Invest 2014; 124(7):2877-90. doi: 10.1172/JCI70156.
doi: 10.1172/JCI70156 pmid: 24865425 |
| 86. |
Ying J, Li H, Chen YW, et al. WNT5A is epigenetically silenced in hematologic malignancies and inhibits leukemia cell growth as a tumor suppressor. Blood 2007; 110(12):4130-1. doi: 10.1182/blood-2007-06-094870.
doi: 10.1182/blood-2007-06-094870 pmid: 18024799 |
| 87. |
Hasan MK, Yu J, Chen L, et al. Wnt5a induces ROR1 to complex with HS1 to enhance migration of chronic lymphocytic leukemia cells. Leukemia 2017; 31(12):2615-22. doi: 10.1038/leu.2017.133.
pmid: 28465529 |
| 88. |
Despeaux M, Chicanne G, Rouer E, et al. Focal adhesion kinase splice variants maintain primitive acute myeloid leukemia cells through altered Wnt signaling. Stem Cells 2012; 30(8):1597-610. doi: 10.1002/stem.1157.
doi: 10.1002/stem.1157 pmid: 22714993 |
| 89. |
Yu J, Chen L, Cui B, et al. Wnt5a induces ROR1/ROR2 heterooligomerization to enhance leukemia chemotaxis and proliferation. J Clin Invest 2016; 126(2):585-98. doi: 10.1172/JCI83535.
doi: 10.1172/JCI83535 pmid: 26690702 |
| 90. |
Ng ÖH, Fırtına S, Can İ, et al. A possible role for WNT5A hypermethylation in pediatric acute lymphoblastic leukemia. Turk J Haematol 2015; 32(2):127-35. doi: 10.4274/Tjh.2013.0296.
doi: 10.4274/tjh.2013.0296 pmid: 26316480 |
| 91. | Zhou HR, Fu HY, Wu DS, et al. Relationship between epigenetic changes in Wnt antagonists and acute leukemia. Oncology Rep 2017; 37(5):2663-71. doi: 10.3892/or.2017.5509. |
| [1] | Xu Chen, Hongling Liu, Dehong Li, Jinsui Wang, Fenghui Zhao. Dual Role of Wnt5a in the Progression of Inflammatory Diseases [J]. Chinese Medical Sciences Journal, 2022, 37(3): 265-274. |
| [2] | Chun Wang, Qinxue Chang, Xiaomeng Wang, Keyun Wang, He Wang, Zhuang Cui, Changping Li. Prostate Cancer Risk Prediction and Online Calculation Based on Machine Learning Algorithm [J]. Chinese Medical Sciences Journal, 2022, 37(3): 210-217. |
| [3] | Siwen Ouyang, Weiming Kang. Research Advances in the Role of Keratins in Gastrointestinal Cancer [J]. Chinese Medical Sciences Journal, 2022, 37(1): 73-78. |
| [4] | Liang Wang, Gang Li, Yun-tao Bing, Mao-lin Tian, Hangyan Wang, Chunhui Yuan, Dianrong Xiu. Does Prior Cancer Have an Influence on the Survival Outcomes of Patients with Localized Pancreatic Neuroendocrine Tumors? [J]. Chinese Medical Sciences Journal, 2021, 36(4): 284-294. |
| [5] | Wei Yang, Mei Zhang. Biomarkers with Potential Predictive Value for Cardiotoxicity in Anticancer Treatments [J]. Chinese Medical Sciences Journal, 2021, 36(4): 333-341. |
| [6] | Chen Xu, Huo Xiaofei, Wu Zhe, Lu Jingjing. Advances of Artificial Intelligence Application in Medical Imaging of Ovarian Cancers [J]. Chinese Medical Sciences Journal, 2021, 36(3): 196-203. |
| [7] | Xiaoqi Zeng, Shanshan Jiang, Yuanyang Peng, Minfeng Liu, Changsheng Ye, Jianyu Dong. Trastuzumab-Induced Severe Thrombocytopenia:A Case Report and Literature Review [J]. Chinese Medical Sciences Journal, 2020, 35(4): 377-382. |
| [8] | Li Wenxing, Zhang Yanli. Novel Long Non-coding RNA Markers for Prognostic Prediction of Patients with Bladder Cancer [J]. Chinese Medical Sciences Journal, 2020, 35(3): 239-247. |
| [9] | Wang Zhan, Wang Xu, Wang Wenda, Zheng Guoyang, Guo Hao, Zhang Yushi. Value of Preoperative Neutrophil-to-Lymphocyte Ratio in Predicting Prognosis of Surgically Resectable Urinary Cancers: Systematic Review and Meta-Analysis [J]. Chinese Medical Sciences Journal, 2020, 35(3): 262-271. |
| [10] | Xu Longming,Dai Shuiping,Zuo Yunxia. Impacts of Preoperative Smoking and Smoking Cessation Time on Preoperative Peripheral Blood Inflammatory Indexes and Postoperative Hospitalization Outcome in Male Patients with Lung Cancer and Surgery Treatment [J]. Chinese Medical Sciences Journal, 2020, 35(2): 170-178. |
| [11] | Zhang Ling, Sun Lei, Mu Xiaoyan, Ji Youxin. Bevacizumab Combined with Icotinib Overcomes Osimertinib Resistance in a Patient of Non-Small Cell Lung Cancer [J]. Chinese Medical Sciences Journal, 2019, 34(4): 292-296. |
| [12] | Liang Xi, Hu Jingnan, He Jianming. An Optimized Protocol of Azoxymethane-Dextran Sodium Sulfate Induced Colorectal Tumor Model in Mice [J]. Chinese Medical Sciences Journal, 2019, 34(4): 281-288. |
| [13] | Bao Yi, Mo Juanfen, Wu Jiayuan, Cao Chenxi. Expression of PD1 and BTLA on the CD8+ T Cell and γδT Cell Subsets in Peripheral Blood of Non-Small Cell Lung Cancer Patients [J]. Chinese Medical Sciences Journal, 2019, 34(4): 248-255. |
| [14] | Wang Guorong, Wang Zhiwei, Jin Zhengyu. Application and Progress of Texture Analysis in the Therapeutic Effect Prediction and Prognosis of Neoadjuvant Chemoradiotherapy for Colorectal Cancer [J]. Chinese Medical Sciences Journal, 2019, 34(1): 45-50. |
| [15] | Liu Qian, Ning Xiaohong, Wang Lei, Liu Wei. Individualized Aromatherapy in End-of-Life Cancer Patients Care: A Case Report [J]. Chinese Medical Sciences Journal, 2018, 33(4): 234-239. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
|