Chinese Medical Sciences Journal ›› 2023, Vol. 38 ›› Issue (1): 49-56.doi: 10.24920/004137
原发性醛固酮增多症的药物治疗
- 中国医学科学院北京协和医院泌尿外科,北京100730,中国
-
收稿日期:2022-07-01接受日期:2022-09-23出版日期:2023-03-31发布日期:2023-01-16 -
通讯作者:文进 E-mail:wjpumch@163.com.
Advances in Medical Treatment of Primary Aldosteronism
Ying-Jie Li,Zhi-Gang Ji,Jin Wen*( )
)
- Department of Urology, Peking Union Medical College Hospital, Chinese Academy of Medical Science & Peking Union Medical College, Beijing 100730, China
-
Received:2022-07-01Accepted:2022-09-23Published:2023-03-31Online:2023-01-16 -
Contact:Jin Wen E-mail:wjpumch@163.com.
摘要:
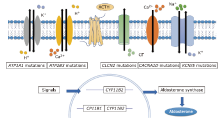
原发性醛固酮增多症(primary aldosteronism,PA)是继发性高血压最常见的形式,主要表现为高血压和低钾血症。原发性醛固酮增多症的早期识别是非常重要的,因为原发性醛固酮增多症患者发生心血管并发症的可能性更高,如房颤、卒中和心肌梗死。在过去的十年中,原发性醛固酮增多症的遗传学研究取得了惊人的进展,为其治疗提供了新的指导。单侧病变首选手术治疗,双侧病变可用盐皮质激素受体拮抗剂(mineralocorticoid receptor antagonist,MRA)治疗。新一代非甾体MRA正在研究中。新的药物治疗策略包括钙通道阻滞剂、大环内酯类抗生素、醛固酮合成酶抑制剂,为原发性醛固酮增多症的治疗提供了新的视角。
引用本文
Ying-Jie Li, Zhi-Gang Ji, Jin Wen. Advances in Medical Treatment of Primary Aldosteronism[J].Chinese Medical Sciences Journal, 2023, 38(1): 49-56.
| [1] |
Brown JM, Siddiqui M, Calhoun DA, et al. The unrecognized prevalence of primary aldosteronism: a cross-sectional study. Ann Internal Med 2020; 173(1):10-20. doi: 10.7326/M20-0065.
doi: 10.7326/M20-0065 |
| [2] |
Choy KW, Fuller PJ, Russell G, et al. Primary aldosteronism. BMJ (Clinical research ed) 2022; 377: e065250. doi:10.1136/bmj-2021-065250.
doi: 10.1136/bmj-2021-065250 |
| [3] |
Zennaro MC, Boulkroun S, Fernandes-Rosa FL. Pathogenesis and treatment of primary aldosteronism. Nat Rev Endocrinol 2020; 16: 578-89. doi:10.1038/s41574-020-0382-4.
doi: 10.1038/s41574-020-0382-4 |
| [4] |
Reincke M, Bancos I, Mulatero P, et al. Diagnosis and treatment of primary aldosteronism. Lancet Diabetes & endocrinology 2021; 9: 876-92. doi:10.1016/s2213-8587(21)00210-2.
doi: 10.1016/s2213-8587(21)00210-2 |
| [5] |
Rossi GP. Primary aldosteronism: Jacc state-of-the-art review. J Am Coll Cardiol 2019; 74: 2799-811. doi:10.1016/j.jacc.2019.09.057.
doi: S0735-1097(19)37927-6 pmid: 31779795 |
| [6] |
Fernandes-Rosa FL, Boulkroun S, Zennaro MC. Genetic and genomic mechanisms of primary aldosteronism. Trends Mol Med 2020; 26: 819-32. doi:10.1016/j.molmed.2020.05.005.
doi: S1471-4914(20)30134-9 pmid: 32563556 |
| [7] |
Santana LS, Guimaraes AG, Almeida MQ. Pathogenesis of primary aldosteronism: impact on clinical outcome. Front Endocrinol (Lausanne) 2022; 13: 927669. doi:10.3389/fendo.2022.927669.
doi: 10.3389/fendo.2022.927669 |
| [8] |
Kitamoto T, Suematsu S, Yamazaki Y, et al. Clinical and steroidogenic characteristics of aldosterone-producing adenomas with atpase or cacna1d gene mutations. J Clin Endocrinol Metab 2016; 101: 494-503. doi:10.1210/jc.2015-3284.
doi: 10.1210/jc.2015-3284 pmid: 26606680 |
| [9] |
Lifton RP, Dluhy RG, Powers M, et al. A chimaeric 11 beta-hydroxylase/aldosterone synthase gene causes glucocorticoid-remediable aldosteronism and human hypertension. Nature 1992; 355: 262-65. doi:10.1038/355262a0.
doi: 10.1038/355262a0 |
| [10] |
Scholl UI, Stölting G, Schewe J, et al. Clcn2 chloride channel mutations in familial hyperaldosteronism type ii. Nat Genet 2018; 5: 349-54. doi:10.1038/s41588-018-0048-5.
doi: 10.1038/s41588-018-0048-5 |
| [11] |
Choi M, Scholl UI, Yue P, et al. K+ channel mutations in adrenal aldosterone-producing adenomas and hereditary hypertension. Science (New York, NY) 2011; 331: 768-72. doi:10.1126/science.1198785.
doi: 10.1126/science.1198785 |
| [12] |
Fernandes-Rosa FL, Daniil G, Orozco IJ, et al. A gain-of-function mutation in the clcn2 chloride channel gene causes primary aldosteronism. Nat Genet 2018; 50: 355-61. doi:10.1038/s41588-018-0053-8.
doi: 10.1038/s41588-018-0053-8 pmid: 29403012 |
| [13] |
Nanba K, Omata K, Else T, et al. Targeted molecular characterization of aldosterone-producing adenomas in white americans. J Clin Endocrinol Metab 2018; 103: 3869-76. doi:10.1210/jc.2018-01004.
doi: 10.1210/jc.2018-01004 pmid: 30085035 |
| [14] |
De Sousa K, Boulkroun S, Baron S, et al. Genetic, cellular, and molecular heterogeneity in adrenals with aldosterone-producing adenoma. Hypertension 2020; 75: 1034-44. doi:10.1161/hypertensionaha.119.14177.
doi: 10.1161/HYPERTENSIONAHA.119.14177 pmid: 32114847 |
| [15] |
Scholl UI, Goh G, Stölting G, et al. Somatic and germline cacna1d calcium channel mutations in aldosterone-producing adenomas and primary aldosteronism. Nat Genet 2013; 45: 1050-54. doi:10.1038/ng.2695.
doi: 10.1038/ng.2695 pmid: 23913001 |
| [16] |
Young WF Jr. Diagnosis and treatment of primary aldosteronism: Practical clinical perspectives. J Intern Med 2019; 285: 126-48. doi:10.1111/joim.12831.
doi: 10.1111/joim.12831 pmid: 30255616 |
| [17] |
Tauber P, Aichinger B, Christ C, et al. Cellular pathophysiology of an adrenal adenoma-associated mutant of the plasma membrane ca(2+)-atpase atp2b3. Endocrinology 2016; 157: 2489-99. doi:10.1210/en.2015-2029.
doi: 10.1210/en.2015-2029 pmid: 27035656 |
| [18] |
Dutta RK, Arnesen T, Heie A, et al. A somatic mutation in clcn2 identified in a sporadic aldosterone-producing adenoma. Eur J Endocrinol 2019; 181: K37-41. doi:10.1530/eje-19-0377.
doi: 10.1530/eje-19-0377 |
| [19] |
Williams TA, Lenders JWM, Mulatero P, et al. Outcomes after adrenalectomy for unilateral primary aldosteronism: an international consensus on outcome measures and analysis of remission rates in an international cohort. Lancet Diabetes Endocrinol 2017; 5: 689-99. doi:10.1016/s2213-8587(17)30135-3.
doi: 10.1016/s2213-8587(17)30135-3 |
| [20] |
Lechner B, Lechner K, Heinrich D, et al. Therapy of endocrine disease: Medical treatment of primary aldosteronism. European J Endocrinol 2019; 181: R147-53. doi:10.1530/eje-19-0215.
doi: 10.1530/eje-19-0215 |
| [21] |
Ahmed S, Hundemer GL. Benefits of surgical over medical treatment for unilateral primary aldosteronism. Front Endocrinol (Lausanne) 2022; 13: 861581. doi:10.3389/fendo.2022.861581.
doi: 10.3389/fendo.2022.861581 |
| [22] |
El-Asmar N, Rajpal A, Arafah BM. Primary hyperaldosteronism: Approach to diagnosis and management. Med Clin N Am 2021; 105: 1065-80. doi:10.1016/j.mcna.2021.06.007.
doi: 10.1016/j.mcna.2021.06.007 |
| [23] |
Pan CT, Liao CW, Tsai CH, et al. Influence of different treatment strategies on new-onset atrial fibrillation among patients with primary aldosteronism: a nationwide longitudinal cohort-based study. J Am Heart Assoc 2020; 9: e013699. doi:10.1161/jaha.119.013699.
doi: 10.1161/jaha.119.013699 |
| [24] |
Velema M, Dekkers T, Hermus A, et al. Quality of life in primary aldosteronism: A comparative effectiveness study of adrenalectomy and medical treatment. J Clin Endocrinol Metab 2018; 103: 16-24. doi:10.1210/jc.2017-01442.
doi: 10.1210/jc.2017-01442 pmid: 29099925 |
| [25] |
Parthasarathy HK, Ménard J, White WB, et al. A double-blind, randomized study comparing the antihypertensive effect of eplerenone and spironolactone in patients with hypertension and evidence of primary aldosteronism. J Hypertens 2011; 29: 980-90. doi:10.1097/HJH.0b013e3283455ca5.
doi: 10.1097/HJH.0b013e3283455ca5 pmid: 21451421 |
| [26] |
Karashima S, Yoneda T, Kometani M, et al. Comparison of eplerenone and spironolactone for the treatment of primary aldosteronism. Hypertens Res 2016; 39: 133-7. doi:10.1038/hr.2015.129.
doi: 10.1038/hr.2015.129 pmid: 26606875 |
| [27] |
Reincke M, Fischer E, Gerum S, et al. Observational study mortality in treated primary aldosteronism: The german conn's registry. Hypertension 2012; 60: 618-24. doi:10.1161/hypertensionaha.112.197111.
doi: 10.1161/HYPERTENSIONAHA.112.197111 pmid: 22824982 |
| [28] |
Catena C, Colussi G, Lapenna R, et al. Long-term cardiac effects of adrenalectomy or mineralocorticoid antagonists in patients with primary aldosteronism. Hypertension 2007; 50: 911-8. doi:10.1161/hypertensionaha.107.095448.
doi: 10.1161/HYPERTENSIONAHA.107.095448 pmid: 17893375 |
| [29] |
Indra T, Holaj R, Štrauch B, et al. Long-term effects of adrenalectomy or spironolactone on blood pressure control and regression of left ventricle hypertrophy in patients with primary aldosteronism. J Renin Angiotensin Aldosterone Syst 2015; 16: 1109-17. doi:10.1177/1470320314549220.
doi: 10.1177/1470320314549220 |
| [30] |
Hundemer GL, Curhan GC, Yozamp N, et al. Cardiometabolic outcomes and mortality in medically treated primary aldosteronism: A retrospective cohort study. Lancet Diabetes Endocrinol 2018; 6: 51-9. doi:10.1016/s2213-8587(17)30367-4.
doi: 10.1016/s2213-8587(17)30367-4 |
| [31] |
Hundemer GL, Curhan GC, Yozamp N, et al. Incidence of atrial fibrillation and mineralocorticoid receptor activity in patients with medically and surgically treated primary aldosteronism. JAMA Cardiol 2018; 3: 768-74. doi:10.1001/jamacardio.2018.2003.
doi: 10.1001/jamacardio.2018.2003 pmid: 30027227 |
| [32] |
Kolkhof P, Bärfacker L. 30 years of the mineralocorticoid receptor: Mineralocorticoid receptor antagonists: 60 years of research and development. J Endocrinol 2017; 234: T125-40. doi:10.1530/joe-16-0600.
doi: 10.1530/joe-16-0600 |
| [33] |
Pandey AK, Bhatt DL, Cosentino F, et al. Non-steroidal mineralocorticoid receptor antagonists in cardiorenal disease. Eur Heart J 2022; 43(31):2931-45. doi: 10.1093/eurheartj/ehac299.
doi: 10.1093/eurheartj/ehac299 pmid: 35713973 |
| [34] |
Bramlage P, Swift SL, Thoenes M, et al. Non-steroidal mineralocorticoid receptor antagonism for the treatment of cardiovascular and renal disease. Eur J Heart Fail 2017; 19: 811. doi:10.1002/ejhf.888.
doi: 10.1002/ejhf.888 pmid: 28586538 |
| [35] |
Bakris GL, Agarwal R, Anker SD, et al. Effect of finerenone on chronic kidney disease outcomes in type 2 diabetes. N Engl J Med 2020; 383: 2219-29. doi:10.1056/NEJMoa2025845.
doi: 10.1056/NEJMoa2025845 |
| [36] |
Motomura N, Yamazaki Y, Gao X, et al. Visualization of calcium channel blockers in human adrenal tissues and their possible effects on steroidogenesis in the patients with primary aldosteronism (pa). J Steroid Biochem 2022; 218: 106062. doi:10.1016/j.jsbmb.2022.106062.
doi: 10.1016/j.jsbmb.2022.106062 |
| [37] |
Yang T, He M, Zhang H, et al. L- and t-type calcium channels control aldosterone production from human adrenals. J Endocrinol 2020; 244: 237-47. doi:10.1530/joe-19-0259.
doi: 10.1530/JOE-19-0259 pmid: 31652415 |
| [38] |
Tauber P, Penton D, Stindl J, et al. Pharmacology and pathophysiology of mutated kcnj5 found in adrenal aldosterone-producing adenomas. Endocrinology 2014; 155: 1353-62. doi:10.1210/en.2013-1944.
doi: 10.1210/en.2013-1944 pmid: 24506072 |
| [39] |
Wang F, Ma X, Tong A, et al. The effects of different calcium channel blockers on aldosterone-producing adenoma cells. Front Endocrinol (Lausanne) 2020; 11: 260. doi:10.3389/fendo.2020.00260.
doi: 10.3389/fendo.2020.00260 |
| [40] |
De Mingo Alemany MC, Mifsud Grau L, Moreno Macián F, et al. A de novo cacna1d missense mutation in a patient with congenital hyperinsulinism, primary hyperaldosteronism and hypotonia. Channels (Austin, Tex) 2020; 14: 175-80. doi:10.1080/19336950.2020.1761171.
doi: 10.1080/19336950.2020.1761171 |
| [41] |
Scholl UI, Abriola L, Zhang C, et al. Macrolides selectively inhibit mutant kcnj5 potassium channels that cause aldosterone-producing adenoma. J Clin Invest 2017; 127: 2739-50. doi:10.1172/JCI91733.
doi: 10.1172/JCI91733 pmid: 28604387 |
| [42] |
Caroccia B, Prisco S, Seccia TM, et al. Macrolides blunt aldosterone biosynthesis: A proof-of-concept study in kcnj5 mutated adenoma cells ex vivo. Hypertension 2017; 70: 1238-42. doi:10.1161/HYPERTENSIONAHA.117.10226.
doi: 10.1161/HYPERTENSIONAHA.117.10226 |
| [43] |
Maiolino G, Ceolotto G, Battistel M, et al. Macrolides for kcnj5-mutated aldosterone-producing adenoma (mapa): design of a study for personalized diagnosis of primary aldosteronism. Blood Press 2018; 27: 200-5. doi:10.1080/08037051.2018.1436961.
doi: 10.1080/08037051.2018.1436961 |
| [44] |
Calhoun DA, White WB, Krum H, et al. Effects of a novel aldosterone synthase inhibitor for treatment of primary hypertension: results of a randomized, double-blind, placebo- and active-controlled phase 2 trial. Circulation 2011; 124: 1945-55. doi:10.1161/circulationaha.111.029892.
doi: 10.1161/CIRCULATIONAHA.111.029892 pmid: 21986283 |
| [45] |
Zhang SL, Gao JW, Guo Y, et al. Associations between metabolic profiles and target-organ damage in chinese individuals with primary aldosteronism. Front Endocrinol (Lausanne) 2020; 11: 547356. doi:10.3389/fendo.2020.547356.
doi: 10.3389/fendo.2020.547356 |
| [46] |
Wu J, Ding X, Tan X. A patent review of aldosterone synthase inhibitors (2014-present). Expert opinion on therapeutic patents 2022; 32: 13-28. doi:10.1080/13543776.2021.1965991.
doi: 10.1080/13543776.2021.1965991 |
| [47] |
Weldon SM, Brown NF. Inhibitors of aldosterone synthase. Vitam Horm 2019; 109: 211-39. doi:10.1016/bs.vh.2018.10.002.
doi: S0083-6729(18)30074-8 pmid: 30678857 |
| [48] |
Amar L, Azizi M, Menard J, et al. Sequential comparison of aldosterone synthase inhibition and mineralocorticoid blockade in patients with primary aldosteronism. J Hypertens 2013; 31: 624-29; discussion 629. doi:10.1097/HJH.0b013e32835d6d49.
doi: 10.1097/HJH.0b013e32835d6d49 pmid: 23314743 |
| [49] |
Lenzini L, Zanotti G, Bonchio M, et al. Aldosterone synthase inhibitors for cardiovascular diseases: a comprehensive review of preclinical, clinical and in silico data. Pharmacological research 2021; 163: 105332. doi:10.1016/j.phrs.2020.105332.
doi: 10.1016/j.phrs.2020.105332 |
| [50] |
Bogman K, Schwab D, Delporte ML, et al. Preclinical and early clinical profile of a highly selective and potent oral inhibitor of aldosterone synthase (cyp11b2). Hypertension 2017; 69: 189-1. doi:10.1161/HYPERTENSIONAHA.116.07716.
doi: 10.1161/HYPERTENSIONAHA.116.07716 pmid: 27872236 |
| [51] |
Fu SJ, Hu MC, Peng YJ, et al. Cul4-ddb1-crbn e 3 ubiquitin ligase regulates proteostasis of clc-2 chloride channels: implication for aldosteronism and leukodystrophy. Cells 2020; 9. doi:10.3390/cells9061332.
doi: 10.3390/cells9061332 |
| [52] |
Rossi GP, Lenzini L. Vitamin d supplementation: A novel therapy for aldosteronism? Nat Rev Endocrinol 2020; 16: 303-4. doi:10.1038/s41574-020-0359-3.
doi: 10.1038/s41574-020-0359-3 pmid: 32286517 |
| [53] |
Ismail NA, Kamaruddin NA, Azhar Shah S, et al. The effect of vitamin d treatment on clinical and biochemical outcomes of primary aldosteronism. Clin Endocrinol (Oxf) 2020; 92: 509-17. doi:10.1111/cen.14177.
doi: 10.1111/cen.14177 |
| [54] |
Kumar KG, Trevaskis JL, Lam DD, et al. Identification of adropin as a secreted factor linking dietary macronutrient intake with energy homeostasis and lipid metabolism. Cell metabolism 2008; 8: 468-81. doi:10.1016/j.cmet.2008.10.011.
doi: 10.1016/j.cmet.2008.10.011 pmid: 19041763 |
| [55] |
Stelcer E, Milecka P, Komarowska H, et al. Adropin stimulates proliferation and inhibits adrenocortical steroidogenesis in the human adrenal carcinoma (hac15) cell line. Front Endocrinol (Lausanne) 2020; 11: 561370. doi:10.3389/fendo.2020.561370.
doi: 10.3389/fendo.2020.561370 |
| [56] |
Watanabe D, Morimoto S, Morishima N, et al. Adrenal (pro)renin receptor expression and serum soluble (pro)renin receptor concentration in primary aldosteronism. Int J Endocrinol 2020; 2020: 9640103. doi:10.1155/2020/9640103.
doi: 10.1155/2020/9640103 |
| No related articles found! |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
|