Chinese Medical Sciences Journal ›› 2023, Vol. 38 ›› Issue (2): 77-93.doi: 10.24920/004213
• 指南与共识 • 下一篇
息肉状脉络膜血管病变治疗中国指南(2022)
陈有信1,2,张誉清3,4,5,6,陈长征7,戴虹8,李甦雁9,马翔10,孙晓东11,唐仕波12,王雨生13,魏文斌14,文峰15,徐格致16,于伟泓1,2,张美霞17,赵明威18,张阳19,齐方19,许迅20,*( ),黎晓新21,22,*(
),黎晓新21,22,*( )
)
- 1中国医学科学院北京协和医院眼科
2中国医学科学院北京协和医院眼底病重点实验室
3加拿大安大略省汉密尔顿市麦克马斯特大学健康研究方法/证据/影响中心
4宁波诺丁汉大学GRADE中心
5中国中医科学院针灸研究所
6中国中医科学院广安门医院心血管中西医病症结合数据科学及临床研究中心
7武汉大学附属人民医院眼科
8北京医院眼科
9徐州市第一人民医院眼科
10大连医科大学附属第一医院眼科
11上海市第一人民医院眼科
12长沙爱尔眼科医院
13西安西京医院眼科
14北京同仁医院眼科
15广州中山眼科中心眼科国家重点实验室
16上海复旦大学附属眼耳鼻喉科医院眼科
17四川大学华西医院眼科
18北京大学人民医院眼科
19上海道田循证科技有限公司
20上海市第一人民医院眼科
21厦门大学眼科中心
22北京大学人民医院人民眼科中心
-
接受日期:2023-04-06出版日期:2023-06-30发布日期:2023-05-31
Chinese Guideline on the Management of Polypoidal Choroidal Vasculopathy (2022)
You-Xin Chen1,2,Yu-Qing Zhang3,4,5,6,Chang-Zheng Chen7,Hong Dai8,Su-Yan Li9,Xiang Ma10,Xiao-Dong Sun11,Shi-Bo Tang12,Yu-Sheng Wang13,Wen-Bin Wei14,Feng Wen15,Ge-Zhi Xu16,Wei-Hong Yu1,2,Mei-Xia Zhang17,Ming-Wei Zhao18,Yang Zhang19,Fang Qi19,Xun Xu20,*( ),Xiao-Xin Li21,22,*(
),Xiao-Xin Li21,22,*( )
)
- 1Department of Ophthalmology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China
2Key Laboratory of Ocular Fundus Diseases, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China
3Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, ON, Canada
4Ningbo Nottingham GRADE center, University of Nottingham, Ningbo, China
5Institute of Acupuncture and Moxibustion, China Academy of Chinese Medical Sciences, Beijing, China
6CEBIM (Center for Evidence Based Integrative Medicine)-Clarity Collaboration, Guang'anmen Hospital, China Academy of Chinese Medical Sciences, Beijing, China
7Department of Ophthalmology, Renmin Hospital of Wuhan University, Wuhan, China
8Department of Ophthalmology, Beijing Hospital, Beijing, China
9Department of Ophthalmology, Xuzhou Municipal Hospital Affiliated to Xuzhou medical University, Xuzhou, China
10Department of Ophthalmology, the First Affiliated Hospital of Dalian Medical University, Dalian, China
11Department of Ophthalmology, Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
12Aire Eye Hospital, Changsha, China
13Department of Ophthalmology, Xijing Hospital, Fouth Military Medical University, Xi‘an, China
14Department of Ophthalmology, Beijing Tongren Hospital, Beijing, China
15State Key Laboratory of Ophthalmology, Zhongshan Ophthalmic Center, Sun Yat-sen University, Guangzhou, China
16Ophthalmology, the Affiliated Eye and ENT Hospital, Shanghai Medical School, Fudan University, Shanghai, China
17Department of Ophthalmology, West China School of Medicine, West China Hospital, Sichuan University, Chengdu, China
18Department of Ophthalmology, Peking University People’s Hospital, Beijing, China
19Department of Ophthalmology, Peking University People’s Hospital, Beijing, China
20Department of Ophthalmology, Shanghai General Hospital, Shanghai, China
21Eye Center of Xiamen University, Xiamen, China
22People Eye Center of People’s Hospital, Peking University, Beijing, China
-
Accepted:2023-04-06Published:2023-06-30Online:2023-05-31 -
Contact:*Xun Xu, E-mail:drxuxun@sjtu.edu.cn ; Xiaoxin Li, E-mail:dr_lixiaoxin@163.com
摘要:
背景 在中国,新生血管性年龄相关黄斑变性患者中约有40%患有多灶性脉络膜血管病变(polypoidal choroidal vasculopathy,PCV)。PCV会导致反复视网膜色素上皮脱离(pigment epithelium detachment, PED)、广泛的视网膜下或玻璃体内出血,以及严重的视力损失。近年来,已有多种该病的治疗方法在中国使用,临床医生需要获得治疗PCV的全面经验。
方法 由14名具有专业背景的眼科专家形成指南专家组,重点关注如下方面问题和相应的结局:非活动性PCV患者的治疗选择;未经治疗的PCV患者的治疗选择,包括抗血管内皮生长因子(vascular endothelial growth factor,VEGF)单药治疗、光动力疗法(photodynamic therapy,PDT)单药治疗或联合治疗;抗VEGF负荷治疗后仍有持续性视网膜下液(subretinal fluid,SRF)或视网膜内液(intraretinal fluid,IRF)的患者,以及伴有大量视网膜下出血的患者的治疗选择。证据整合团队在各个问题下进行系统评价,为推荐意见的形成提供依据。本指南采用GRADE方法评估证据的可信性,并确定推荐的强度。
结果 指南专家组共形成了以下六条弱强度推荐意见:(1)对于非活动性PCV患者,建议观察而非立即治疗;(2)对于未经治疗的PCV患者,建议采用抗VEGF单药治疗或抗VEGF联合PDT,而非单独使用PDT;(3)对于拟进行抗VEGF联合PDT治疗的PCV患者,建议采用延迟/挽救PDT而非起始联合PDT;(4)对于拟进行抗VEGF单药治疗的PCV患者,建议在完成三个月负荷治疗后采用“治疗和延长”(treat and extend,T&E)方案,而非“按需治疗” (pro re nata,PRN)方案;(5)对于抗VEGF负荷治疗三个月后在光学相干断层扫描(optical coherence tomography,OCT)上出现持续性SRF或IRF的患者,建议继续抗VEGF治疗而非停药观察;(6)对于伴有新鲜大量视网膜出血且累及黄斑区的PCV患者,建议进行玻璃体切除术联合玻璃体腔内注射组织纤溶酶原激活剂及气体填充,而非抗VEGF单药治疗。
结论 本指南共形成6条推荐意见,以支持PCV患者的最佳治疗选择。
引用本文
You-Xin Chen, Yu-Qing Zhang, Chang-Zheng Chen, Hong Dai, Su-Yan Li, Xiang Ma, Xiao-Dong Sun, Shi-Bo Tang, Yu-Sheng Wang, Wen-Bin Wei, Feng Wen, Ge-Zhi Xu, Wei-Hong Yu, Mei-Xia Zhang, Ming-Wei Zhao, Yang Zhang, Fang Qi, Xun Xu, Xiao-Xin Li. Chinese Guideline on the Management of Polypoidal Choroidal Vasculopathy (2022)[J].Chinese Medical Sciences Journal, 2023, 38(2): 77-93.
"
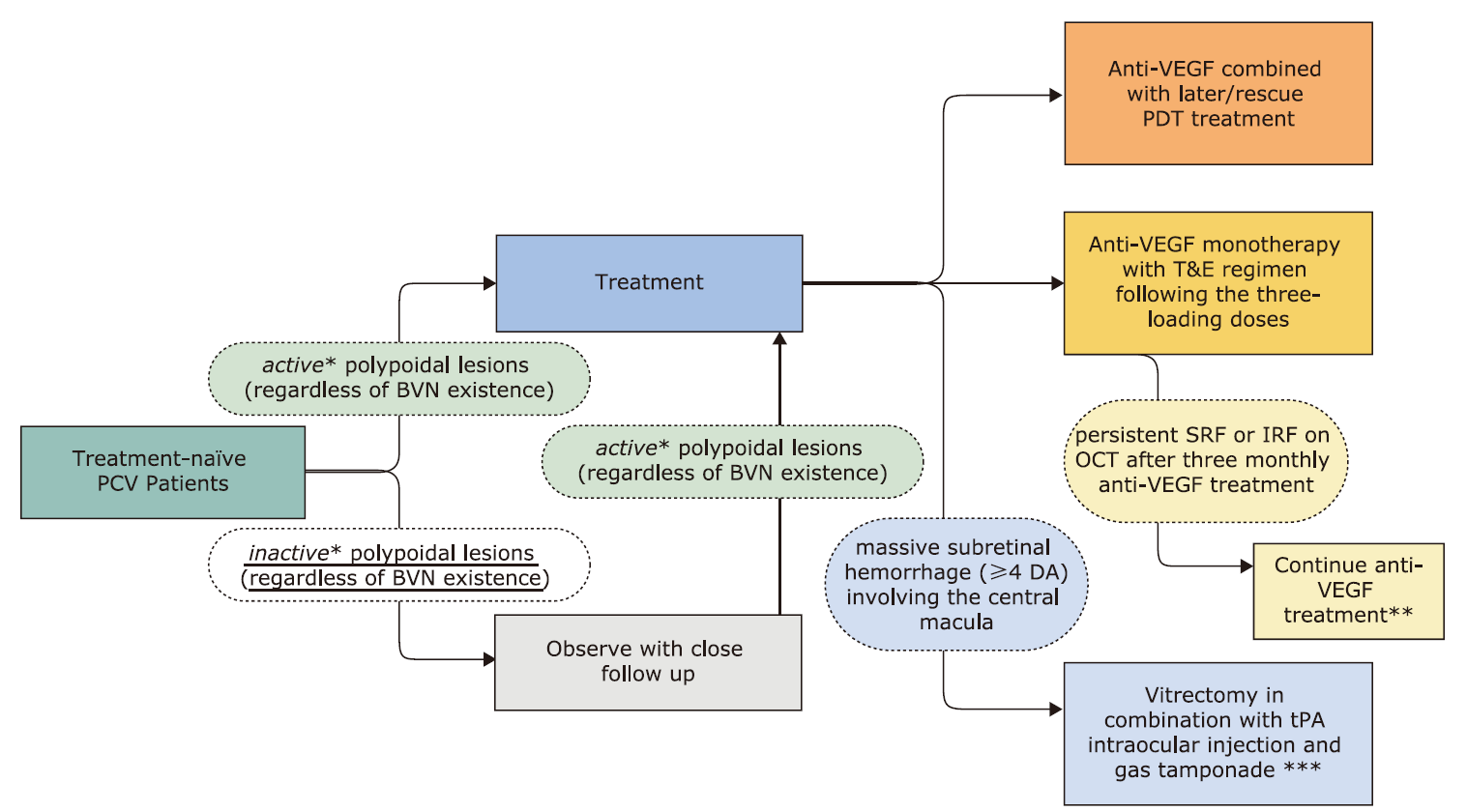
| BOX 1: SUMMARY OF RECOMMENDATIONS | |
|---|---|
| 1 | For treatment-na?ve polypoidal choroidal vasculopathy (PCV) patients with inactive polypoidal lesions, the guideline panel suggests observation over the initiation of treatment (conditional recommendation, very low certainty in the estimated effects). |
| Remarks: Close follow-up and monitoring are essential, especially for patients with high-risk factors (such as cigarette smoking, higher body mass index, and abnormal serum levels of inflammatory markers). | |
| 2 | For treatment-na?ve PCV patients, the guideline panel suggests either anti-VEGF monotherapy or combined anti-VEGF and PDT rather than PDT monotherapy (conditional recommendation, low to very low certainty in the estimated effects). |
| Remarks: The choice may depend on the patient’s condition (such as the size or location of polypoidal lesions and the height of PED) or specific types of anti-VEGF agents. | |
| 3 | For PCV patients who plan to initiate anti-VEGF combined with PDT treatment, the guideline panel suggests later/rescue PDT over initial PDT (conditional recommendation, low certainty in the estimated effects). |
| Remarks: The timing of later PDT may be at least after three months of anti-VEGF according to treatment criteria of PDT (such as if polypoidal lesions are seen with subretinal fluid on the ICGA images obtained) | |
| 4 | For PCV patients who plan to initiate the treatment with anti-VEGF, the guideline panel suggests treat and extend (T&E) over the pro re nata (PRN) regimen following three monthly loading doses (conditional recommendation, very low certainty in the estimated effects). |
| Remarks: The T&E regimen increases the number of injections compared to the PRN regimen, although it reduces the number of visits. The follow-up should consider the morphological changes of the fundus and pay more attention to the functional or conscious symptoms of the affected eye. The interval of T&E can be referred to in the ALTAIR study. | |
| 5 | For PCV patients with persistent subretinal fluid (SRF) or intraretinal fluid (IRF) on optical coherence tomography (OCT) after three monthly anti-VEGF treatments, the guideline panel suggests proceeding with anti-VEGF treatment over observation (conditional recommendation, very low certainty of the estimated effects). |
| Remarks: Clinicians should closely monitor the change in fundus morphology and function of the affected eye (or subjective symptoms) during follow-up and may consider stopping treatment when no clear benefit to visual acuity with further injection is expected, such as extensive subretinal scar formation. | |
| 6 | For PCV patients with massive subretinal hemorrhage (equal or more than four optic disc areas) involving the central macula within the onset of 14 days, the panel suggests vitrectomy in combination with tissue-plasminogen activator (tPA) intraocular injection and gas tamponade over anti-VEGF monotherapy (conditional recommendation, very low certainty in the estimated effects). |
| Remarks: Surgery may also benefit PCV patients with subretinal hemorrhage combined with vitreous hemorrhage; clinicians might consider using complementary therapy (e.g., pneumatic displacement, anti-VEGF, PDT, and tPA). | |
| 1 |
Spaide RF, Jaffe GJ, Sarraf D, et al. Consensus Nomenclature for Reporting Neovascular Age-Related Macular Degeneration Data: Consensus on Neovascular Age-Related Macular Degeneration Nomenclature Study Group. Ophthalmology 2020 ;127(5):616-36. doi: 10.1016/j.ophtha.2019.11.004.
doi: 10.1016/j.ophtha.2019.11.004 |
| 2 |
Spaide RF, Yannuzzi LA, Slakter JS, et al. Indocyanine green videoangiography of idiopathic polypoidal choroidal vasculopathy. Retina 1995 ; 15(2):100-10. doi: 10.1097/00006982-199515020-00003.
doi: 10.1097/00006982-199515020-00003 |
| 3 |
Han LH, Yuan LF, Liang X, et al. Combined therapy versus anti-vascular endothelial growth factor monotherapy for polypoidal choroidal vasculopathy: a meta-analysis. Int J Ophthalmol 2017 ; 10(8):1280-9. doi: 10.18240/ijo.2017.08.16.
doi: 10.18240/ijo.2017.08.16 |
| 4 |
Qu J, Cheng Y, Li X, et al. Efficacy of intravitreal injection of conbercept in polypoidal choroidal vasculopathy: Subgroup analysis of the aurora study. Retina 2016 ; 36(5):926-37. doi: 10.1097/iae.0000000000000875.
doi: 10.1097/iae.0000000000000875 |
| 5 |
Li XX, Zhu Q, Egger A, et al. Two different treatment regimens of ranibizumab 0.5 mg for neovascular age-related macular degeneration with or without polypoidal choroidal vasculopathy in Chinese patients: results from the phase IV, randomized, DRAGON study. Acta Ophthalmol 2021 ; 99(3): e336-e45. doi: 10.1111/aos.14588.
doi: 10.1111/aos.14588 |
| 6 |
Cheung CMG, Yanagi Y, Akiba M, et al. Improved detection and diagnosis of polypoidal choroidal vasculopathy using a combination of optical coherence tomography and optical coherence tomography angiography. Retina 2019 ; 39(9):1655-63. doi: 10.1097/iae.0000000000002228.
doi: 10.1097/iae.0000000000002228 |
| 7 |
Koh AHC, Chen LJ, Chen SJ, et al. Polypoidal choroidal vasculopathy: evidence-based guidelines for clinical diagnosis and treatment. Retina 2013 ; 33(4):686-716. doi: 10.1097/IAE.0b013e3182852446.
doi: 10.1097/IAE.0b013e3182852446 |
| 8 |
Bo Q, Yan Q, Shen M, et al. Appearance of polypoidal lesions in patients with polypoidal choroidal vasculopathy using swept-source optical coherence tomographic angiography. JAMA Ophthalmol 2019 ; 137(6):642-50. doi: 10.1001/jamaophthalmol.2019.0449.
doi: 10.1001/jamaophthalmol.2019.0449 |
| 9 |
De Carlo TE, Kokame GT, Kaneko KN, et al. Sensitivity and specificity of detecting polypoidal choroidal vasculopathy with en face optical coherence tomography and optical coherence tomography angiography. Retina 2019 ; 39(7):1343-52. doi: 10.1097/iae.0000000000002139.
doi: 10.1097/iae.0000000000002139 |
| 10 |
Chen LJ, Cheng CK, Yeung L, et al. Management of polypoidal choroidal vasculopathy: Experts consensus in Taiwan. J Formos Med Assoc 2019 ; 19(2):569-76. doi: 10.1016/j.jfma.2019.04.012.
doi: 10.1016/j.jfma.2019.04.012 |
| 11 |
Cheung CMG, Lai TYY, Ruamviboonsuk P, et al. Polypoidal choroidal vasculopathy: definition, pathogenesis, diagnosis, and management. Ophthalmology 2018 ; 125(5):708-24. doi: 10.1016/j.ophtha.2017.11.019.
doi: 10.1016/j.ophtha.2017.11.019 |
| 12 |
Institute of Medicine US committee on Standards for developing Trustworthy clinical practice Guideline. Graham R, Greenfield S, Steinberg E. Clinical Practice Guidelines We Can Trust. Washington(DC): National Academies Press; 2011. doi: 10.17226/13058.
doi: 10.17226/13058 |
| 13 |
Alonso-Coello P, Oxman AD, Moberg J, et al. GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 2: Clinical practice guidelines. BMJ 2016; 353: i2089. doi: 10.1136/bmj.i2089.
doi: 10.1136/bmj.i2089 |
| 14 |
Guyatt GH, Oxman AD, Kunz R, et al. GRADE Working Group. GRADE guidelines: 7. Rating the quality of evidence-inconsistency. J Clin Epidemiol 2011; 64:1294-302. doi: 10.1016/j.jclinepi.2011.03.017.
doi: 10.1016/j.jclinepi.2011.03.017 |
| 15 |
Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008; 336:924-26. doi: 10.1136/bmj.39489.470347.AD.
doi: 10.1136/bmj.39489.470347.AD |
| 16 |
Schunemann HJ, Mustafa R, Brozek J, et al. GRADE Guidelines: 16. GRADE evidence to decision frameworks for tests in clinical practice and public health. J Clin Epidemiol 2016; 76:89-98. doi: 10.1016/j.jclinepi.2016.01.032.
doi: 10.1016/j.jclinepi.2016.01.032 pmid: 26931285 |
| 17 | Higgins JPT, Deeks JJ, Altman DG. Editors. Special topics in statistics. In: Cochrane Handbook for Systematic Reviews of Interventions version 5.1.0. updated March 2011. The cochrane collaberation 2011. Avail-able from www.handbook.cochrane.org. Accessed: October 17, 2019. |
| 18 |
Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 2010; 25:603-5. doi: 10.1007/s10654-010-9491-z.
doi: 10.1007/s10654-010-9491-z pmid: 20652370 |
| 19 | Schunemann HJ, Oxman AD, Vist GE, et al. 12 Interpreting results and drawing conclusions. In: Cochrane handbook for systematic reviews of interventions (lst ed). New York: Wiley; 2008.p359. |
| 20 |
Balshem H, Helfand M, Schunemann HJ, et al. GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol 2011; 64:401-6. doi: 10.1016/j.jclinepi.2010.07.015.
doi: 10.1016/j.jclinepi.2010.07.015 pmid: 21208779 |
| 21 |
Cheung CM, Laude A, Yeo I, et al. Systemic, ocular and genetic risk factors for age-related macular degeneration and polypoidal choroidal vasculopathy in Singaporeans. Sci Rep 2017; 7:41386. doi: 10.1038/srep41386.
doi: 10.1038/srep41386 pmid: 28120909 |
| 22 |
Fujiwara K, Yasuda M, Hata J, et al. Prevalence and risk factors for polypoidal choroidal vasculopathy in a general Japanese population: the hisayama study. Semin Ophthalmol 2018 ; 33(6):813-19. doi: 10.1080/08820538.2018.1506483.
doi: 10.1080/08820538.2018.1506483 |
| 23 |
Meng Q, Huang L, Sun Y, et al. Effect of high-density lipoprotein metabolic pathway gene variations and risk factors on neovascular age-related macular degeneration and polypoidal choroidal vasculopathy in China. PLoS One 2015 ; 10(12):e0143924. doi: 10.1371/journal.pone.0143924.
doi: 10.1371/journal.pone.0143924 |
| 24 |
Sho K, Takahashi K, Yamada H, et al. Polypoidal choroidal vasculopathy: incidence, demographic features, and clinical characteristics. Archiv Ophthalmol 2003 ; 121(10):1392-96. doi: 10.1001/archopht.121.10.1392.
doi: 10.1001/archopht.121.10.1392 |
| 25 |
Woo SJ, Ahn J, Morrison MA, et al. Analysis of genetic and environmental risk factors and their interactions in korean patients with age-related macular degeneration. PLoS One 2015 ; 10(7): e0132771. doi: 10.1371/journal.pone.0132771.
doi: 10.1371/journal.pone.0132771 |
| 26 |
Kikuchi M, Nakamura M, Ishikawa K, et al. Elevated C-reactive protein levels in patients with polypoidal choroidal vasculopathy and patients with neovascular age-related macular degeneration. Ophthalmology 2007 ; 114(9):1722-27. doi: 10.1016/j.ophtha.2006.12.021.
doi: 10.1016/j.ophtha.2006.12.021 |
| 27 |
Sakurada Y, Nakamura Y, Yoneyama S, et al. Aqueous humor cytokine levels in patients with polypoidal choroidal vasculopathy and neovascular age-related macular degeneration. Ophthalmic Res 2015 ; 53(1):2-7. doi: 10.1159/000365487.
doi: 10.1159/000365487 |
| 28 |
Okubo A, Arimura N, Abematsu N, et al. Predictable signs of benign course of polypoidal choroidal vasculopathy: based upon the long-term observation of non-treated eyes. Acta Ophthalmol 2010; 88:e107-e14. doi: 10.1111/j.1755-3768.2009.01850.x.
doi: 10.1111/j.1755-3768.2009.01850.x |
| 29 |
Zeng R, Zhang X, Li M, et al. Pilot study of inactive polypoidal lesions in polypoidal choroidal vasculopathy. Eur J Ophthalmol 2015 ; 25(3):222-28. doi: 10.5301/ejo.5000532.
doi: 10.5301/ejo.5000532 |
| 30 |
Koh A, Lee W, Chen L, et al. EVEREST study: efficacy and safety of verteporfin photodynamic therapy in combination with ranibizumab or alone versus ranibizumab monotherapy in patients with symptomatic macular polypoidal choroidal vasculopathy. Retina 2012 ; 32(8):1453-64. doi: 10.1097/IAE.0b013e31824f91e8.
doi: 10.1097/IAE.0b013e31824f91e8 |
| 31 |
Oishi A, Kojima H, Mandai M, et al. Comparison of the effect of ranibizumab and verteporfin for polypoidal choroidal vasculopathy: 12-month LAPTOP study results. Am J Ophthalmol 2013 ; 156(4):644-51. doi: 10.1016/j.ajo.2013.05.024.
doi: 10.1016/j.ajo.2013.05.024 |
| 32 | Guo Y. [Photodynamic therapy combined with intravitreal injection of ranibizumab for polypoidal choroidal vasculopathy]. [dissertation] Nanhua University; 2014. |
| 33 |
Zhao J, Ma LB. [Efficacy of intravitreal ranibizumab in combination with photodynamic therapy on polypoid choroidal vasculopathy]. Chin J Ophthalmol 2016 ;26(6):387-90. Chinese. doi: 10.13444/j.cnki.zgzyykzz.2016.06.010.
doi: 10.13444/j.cnki.zgzyykzz.2016.06.010 |
| 34 |
Lai KB, Li Y, Zhou LJ, et al. Comparison of the effects of photodynamic therapy, intravitreal ranibizumab and combination for polypoidal choroidal vasculopathy under 1+PRN regimen. BMC Ophthalmol 2018; 18:9. doi: 10.1186/s12886-018-0801-7.
doi: 10.1186/s12886-018-0801-7 |
| 35 |
Lee WK, Iida T, Ogura Y, et al. Efficacy and safety of intravitreal aflibercept for polypoidal choroidal vasculopathy in the PLANET study: a randomized clinical trial. JAMA Ophthalmol 2018 ; 136(7):786-93. doi: 10.1001/jamaophthalmol.2018.1804.
doi: 10.1001/jamaophthalmol.2018.1804 |
| 36 |
Wong T, Ogura Y, Lee W, et al. Efficacy and Safety of intravitreal aflibercept for polypoidal choroidal vasculopathy: two-year results of the aflibercept in polypoidal choroidal vasculopathy study. Am J Ophthalmol 2019; 204:80-9. doi: 10.1016/j.ajo.2019.02.027.
doi: S0002-9394(19)30086-8 pmid: 30849345 |
| 37 |
Koh A, Lai T, Takahashi K, et al. Efficacy and safety of ranibizumab with or without verteporfin photodynamic therapy for polypoidal choroidal vasculopathy: a randomized clinical trial. JAMA Ophthalmol 2017 ; 135(11):1206-13. doi: 10.1001/jamaophthalmol.2017.4030.
doi: 10.1001/jamaophthalmol.2017.4030 |
| 38 |
Yoon LJ, Young LS, June-Gone K, et al. Intravitreal bevacizumab alone versus in combination with photodynamic therapy for the treatment of neovascular maculopathy in patients aged 50 years or older: 1-year results of a prospective clinical study. Acta Ophthalmol 2012; 90: 61-7. doi: 10.1111/j.1755-3768.2009.01841.x.
doi: 10.1111/j.1755-3768.2009.01841.x pmid: 20337606 |
| 39 |
Li J, Sun JH, Li B, et al. Intravitreal ranibizumab injection combined with photodynamic therapy for polypoidal choroidal vasculopathy. Exp Ther Med 2018 ; 15(2):1546-51. doi: 10.3892/etm.2017.5565.
doi: 10.3892/etm.2017.5565 |
| 40 |
Guo H, Zhao M, Yan B. [Comparison of visual prognosis between vitreous cavity injection of ranibizumab and vitreous cavity injection of ranibizumab combined with photodynamic therapy for polypoid choroidal vasculopathy]. J Clin Med Literat 2016 ; 3(58):11637. Chinese. doi: 10.3877/j.issn.2095-8242.2016.58.142.
doi: 10.3877/j.issn.2095-8242.2016.58.142 |
| 41 |
Huang Z, Ding Q, Yan M, et al. Short-term efficacy of conbercept and ranibizumab for polypoidal choroidal vasculopathy. Retina 2019 ; 39(5):889-95. doi: 10.1097/iae.0000000000002035.
doi: 10.1097/iae.0000000000002035 |
| 42 |
Kitahashi M, Baba T, Sakurai M, et al. Pneumatic displacement with intravitreal bevacizumab for massive submacular hemorrhage due to polypoidal choroidal vasculopathy. Clin Ophthalmol 2014; 8:485-92. doi: 10.2147/opth.s55413.
doi: 10.2147/opth.s55413 |
| 43 |
Gomi F, Oshima Y, Mori R, et al. Initial versus delayed photodynamic therapy in combination with ranibizumab for treatment of polypoidal choroidal vasculopathy. Retina 2015 ; 35(8):1569-76. doi: 10.1097/IAE.0000000000000526.
doi: 10.1097/IAE.0000000000000526 |
| 44 |
Ohji M, Takahashi K, Okada AA, et al. Efficacy and safety of intravitreal aflibercept treat-and-extend regimens in exudative age-related macular degeneration: 52- and 96-week findings from ALTAIR : a randomized controlled trial. Adv Ther 2020; 37(3):1173-87. doi: 10.1007/s12325-020-01236-x.
doi: 10.1007/s12325-020-01236-x pmid: 32016788 |
| 45 |
Eldem BM, Muftuoglu G, Topbas S, et al. A randomized trial to compare the safety and efficacy of two ranibizumab dosing regimens in a Turkish cohort of patients with choroidal neovascularization secondary to AMD. Acta ophthalmol 2015 ; 93(6):e458-64. doi: 10.1111/aos.12540.
doi: 10.1111/aos.12540 |
| 46 |
Aurell S, Sjovall K, Paul A, et al. Better visual outcome at 1 year with antivascular endothelial growth factor treatment according to treat-and-extend compared with pro re nata in eyes with neovascular age-related macular degeneration. Acta Ophthalmol 2019 ; 97(5):519-24. doi: 10.1111/aos.13989.
doi: 10.1111/aos.13989 |
| 47 |
Garweg Justus G, Niderprim Sophie A, Russ Hanna M, et al. Comparison of strategies of treatment with ranibizumab in newly-diagnosed cases of neovascular age-related macular degeneration. J Ocular Pharmac Therapeut 2017 ; 33(10):773-8. doi: 10.1089/jop.2017.0006.
doi: 10.1089/jop.2017.0006 |
| 48 |
Hanemoto T, Hikichi Y, Kikuchi N, et al. The impact of different anti-vascular endothelial growth factor treatment regimens on reducing burden for caregivers and patients with wet age-related macular degeneration in a single-center real-world Japanese setting. PLoS One 2017 ; 12(12): e0189035. doi:10.1371/journal.pone.0189035.
doi: 10.1371/journal.pone.0189035 |
| 49 |
Oubraham H, Cohen SY, Samimi S, et al. Inject and extend dosing versus dosing as needed: a comparative retrospective study of ranibizumab in exudative age-related macular degeneration. Retina 2011 ; 31(1):26-30. doi: 10.1097/IAE.0b013e3181de5609.
doi: 10.1097/IAE.0b013e3181de5609 |
| 50 |
Johnston RL, Carius HJ, Skelly A, et al. A retrospective study of ranibizumab treatment regimens for neovascular age-related macular degeneration (nAMD) in Australia and the United Kingdom. Adv Ther 2017 ; 34(3):703-12. doi: 10.1007/s12325-017-0483-1.
doi: 10.1007/s12325-017-0483-1 |
| 51 |
Hatz K, Prunte C. Treat and Extend versus Pro Re Nata regimens of ranibizumab in neovascular age-related macular degeneration: a comparative 12 Month study. Acta ophthalmol 2017 ; 95(1):e67-e72. doi: 10.1111/aos.13031.
doi: 10.1111/aos.13031 |
| 52 |
Spooner K, Fraser-Bell S, Cozzi M, et al. Macular atrophy in eyes with neovascular age-related macular degeneration treated with vascular endothelial growth factor inhibitors using a treat-and-extend or pro re nata regimen: 4-year results of the MANEX study. Ophthalmology 2020 ; 127(12):1663-73. doi: 10.1016/j.ophtha.2020.06.019.
doi: 10.1016/j.ophtha.2020.06.019 |
| 53 |
Augsburger M, Sarra GM, Imesch P. Treat and extend versus pro re nata regimens of ranibizumab and aflibercept in neovascular age-related macular degeneration: a comparative study. Graefes Arch Clin Exp Ophthalmol 2019 ; 257(9):1889-95. doi: 10.1007/s00417-019-04404-0.
doi: 10.1007/s00417-019-04404-0 |
| 54 |
Saito M, Kano M, Itagaki K, et al. Subfoveal choroidal thickness in polypoidal choroidal vasculopathy after switching to intravitreal aflibercept injection. Jpn J Ophthalmol 2015 ; 60(1):35-41. doi: 10.1007/s10384-015-0411-3.
doi: 10.1007/s10384-015-0411-3 |
| 55 |
Moon DRC, Lee DK, Kim SH, et al. Aflibercept treatment for neovascular age-related macular degeneration and polypoidal choroidal vasculopathy refractory to anti-vascular endothelial growth factor. Korean J Ophthalmol 2015 ; 29(4):226-32. doi: 10.3341/kjo.2015.29.4.226.
doi: 10.3341/kjo.2015.29.4.226 |
| 56 |
Azuma K, Obata R, Nomura Y, et al. Angiographic findings of ranibizumab-resistant polypoidal choroidal vasculopathy after switching to a treat-and-extend regimen with intravitreal aflibercept. Retina 2016 ; 36(11):2158-65. doi: 10.1097/IAE.0000000000001047.
doi: 10.1097/IAE.0000000000001047 |
| 57 |
Uyama M, Wada M, Nagai Y, et al. Polypoidal choroidal vasculopathy: natural history. Am J Ophthalmol 2002 ; 133(5):639-48. doi: 10.1016/s0002-9394(02)01404-6.
doi: 10.1016/s0002-9394(02)01404-6 |
| 58 |
Shin JY, Lee JM, Byeon SH. Anti-vascular endothelial growth factor with or without pneumatic displacement for submacular hemorrhage. Am J Ophthalmol 2015 ;159(5):904-14.e1. doi: 10.1016/j.ajo.2015.01.024.
doi: 10.1016/j.ajo.2015.01.024 |
| 59 | Nam K, Kim JY, Lee SJ, et al. Comparison of visual outcome between intravitreal gas injection with t-PA and intravitreal anti-VEGF injection as an initial treatment for submacular hemorrhage associated with ARMD. Invest Ophthalmol Vis Sci 2019 ;60(9):1176. |
| 60 |
Cho HJ, Koh KM, Kim JH, et al. Intravitreal ranibizumab injections with and without pneumatic displacement for treating submacular hemorrhage secondary to neovascular age-related macular degeneration. Retina 2015 ; 35(2):205-12. doi: 10.1097/iae.0000000000000295.
doi: 10.1097/iae.0000000000000295 |
| 61 |
Kang HG, Kang H, Byeon SH, et al. Long-term visual outcomes for treatment of submacular haemorrhage secondary to polypoidal choroidal vasculopathy. Clin Exp Ophthalmol 2018 ; 46(8):916-25. doi: 10.1111/ceo.13198.
doi: 10.1111/ceo.13198 |
| 62 |
Sniatecki JJ, Ho-Yen G, Clarke B, et al. Treatment of submacular hemorrhage with tissue plasminogen activator and pneumatic displacement in age-related macular degeneration. Eur J Ophthalmol 2019;1120672119891625. doi: 10.1177/1120672119891625.
doi: 10.1177/1120672119891625 |
| 63 |
Cho HJ, Koh KM, Kim HS, et al. Anti-vascular endothelial growth factor monotherapy in the treatment of submacular hemorrhage secondary to polypoidal choroidal vasculopathy. Am J Ophthalmol 2013 ; 156(3):524-31. e1. doi: 10.1016/j.ajo.2013.04.029.
doi: 10.1016/j.ajo.2013.04.029 |
| 64 |
Isizaki E, Morishita S, Sato T, et al. Treatment of massive subretinal hematoma associated with age-related macular degeneration using vitrectomy with intentional giant tear. Int Ophthalmol 2016 ; 36(2):199-206. doi: 10.1007/s10792-015-0102-6.
doi: 10.1007/s10792-015-0102-6 |
| 65 |
Jung JH, Lee JK, Lee JE, et al. Results of vitrectomy for breakthrough vitreous hemorrhage associated with age-related macular degeneration and polypoidal choroidal vasculopathy. Retina 2010 ; 30(6):865-73. doi: 10.1097/IAE.0b013e3181c969e9.
doi: 10.1097/IAE.0b013e3181c969e9 |
| 66 |
Kim JH, Kim CG, Lee DW, et al. Intravitreal aflibercept for submacular hemorrhage secondary to neovascular age-related macular degeneration and polypoidal choroidal vasculopathy. Graefes Arch Clin Exp Ophthalmol 2020 ; 258(1):107-16. doi: 10.1007/s00417-019-04474-0.
doi: 10.1007/s00417-019-04474-0 |
| 67 |
Kim KH, Kim JH, Chang YS, et al. Clinical outcomes of eyes with submacular hemorrhage secondary to age-related macular degeneration treated with anti-vascular endothelial growth factor. Korean J Ophthalmol 2015 ; 29(5):315-24. doi: 10.3341/kjo.2015.29.5.315.
doi: 10.3341/kjo.2015.29.5.315 |
| 68 |
Kimura S, Morizane Y, Hosokawa MM, et al. Outcomes of vitrectomy combined with subretinal tissue plasminogen activator injection for submacular hemorrhage associated with polypoidal choroidal vasculopathy. Jpn J Ophthalmol 2019 ; 63(5):382-8. doi: 10.1007/s10384-019-00679-2.
doi: 10.1007/s10384-019-00679-2 |
| 69 |
Kimura S, Morizane Y, Matoba R, et al. Retinal sensitivity after displacement of submacular hemorrhage due to polypoidal choroidal vasculopathy: effectiveness and safety of subretinal tissue plasminogen activator. Jpn J Ophthalmol 2017; 61:472-8. doi: 10.1007/s10384-017-0530-0.
doi: 10.1007/s10384-017-0530-0 pmid: 28836011 |
| 70 |
Kitagawa Y, Shimada H, Mori R, et al. Intravitreal tissue plasminogen activator, ranibizumab, and gas injection for submacular hemorrhage in polypoidal choroidal vasculopathy. Ophthalmology 2016 ; 123(6):1278-86. doi: 10.1016/j.ophtha.2016.01.035.
doi: 10.1016/j.ophtha.2016.01.035 |
| 71 |
Kung YH, Wu TT, Hong MC, et al. Intravitreal tissue plasminogen activator and pneumatic displacement of submacular hemorrhage. J Ocul Pharmacol Ther 2010 ; 26(5):469-74. doi: 10.1089/jop.2010.0066.
doi: 10.1089/jop.2010.0066 |
| 72 |
Lin HC, Yang CH, Yang CM, et al. Visual outcomes of vitrectomy for polypoidal choroidal vasculopathy-related breakthrough vitreous haemorrhage. Eye 2014 ; 28(7):797-806. doi: 10.1038/eye.2014.124.
doi: 10.1038/eye.2014.124 |
| 73 |
Lin TC, Hwang DK, Lee FL, et al. Visual prognosis of massive submacular hemorrhage in polypoidal choroidal vasculopathy with or without combination treatment. J Chin Med Assoc 2016 ; 79(3):159-65. doi: 10.1016/j.jcma.2015.11.004.
doi: 10.1016/j.jcma.2015.11.004 |
| 74 |
Stifter E, Michels S, Prager F, et al. Intravitreal bevacizumab therapy for neovascular age-related macular degeneration with large submacular hemorrhage. Am J Ophthalmologica 2007 ; 144(6):886-92. doi: 10.1016/j.ajo.2007.07.034.
doi: 10.1016/j.ajo.2007.07.034 |
| 75 |
Yang Y, Li J, Liang J, et al. [Observation of polypoidal choroidal vasculopathy with vitreous hemorrhage treated by microincision 25G pars plana vitrectomy]. Int Eye Sci 2020 ; 20(3):571-5. Chinese. doi: 10.3980/j.issn.1672-5123.2020.3.39.
doi: 10.3980/j.issn.1672-5123.2020.3.39 |
| 76 |
Baek J, Kim JH, Lee MY, et al. Disease activity after development of large subretinal hemorrhage in polypoidal choroidal vasculopathy. Retina 2018 ; 38(10):1993-2000. doi: 10.1097/iae.0000000000001817.
doi: 10.1097/iae.0000000000001817 |
| 77 |
Bi X, Chen S, Wang Y, et al. [Observation of vitrectomy on polypoidal choroidal vasculopathy with vitreous hemorrhage]. Chin J Practic Ophthalmol 2012; (6):653-6. Chinese. doi: 10.3760/cma.j.issn.1006-4443.2012.06.005.
doi: 10.3760/cma.j.issn.1006-4443.2012.06.005 |
| 78 |
Li YS, Gao RY, Chen H. [Effect of vitrectomy combined with intravitreal Conbercept injection for polypoidal choroidal vasculopathy associated with vitreous hemorrhage]. Int Eye Sci 2017 ;17(1):113-7. Chinese. doi: 10.3980/j.issn.1672-5123.2017.1.30.
doi: 10.3980/j.issn.1672-5123.2017.1.30 |
| 79 | Pang L. [Efficacy of vitrectomy combined with postoperative anti-VEGF drugs in the treatment of PCV combined with vitreous hemorrhage][dissertation]. Nanchang: Nanchang University; 2016. |
| 80 |
She JT, Zhang GM, Zhao TY, et al. [Influence of intravitreal ranibizumab in pars plana vitrectomy on polypoidal choroidal vasculopathy with vitreous hemorrhage]. Chin J Practic Ophthalmol 2015 ; 33(8):913-17. Chinese. doi: 10.3760/cma.j.issn.1006-4443.2015.08.019.
doi: 10.3760/cma.j.issn.1006-4443.2015.08.019 |
| 81 |
Shen J, Xy LP, Meng XJ, et al. [Effect of vitrectomy with intravitreal Conbercept injection for polypoidal choroidal vasculopathy associated with vitreous hemorrhage]. Int Eye Sci 2018 ;18(9):1660-4. Chinese. doi: 10.3980/j.issn.1672-5123.2018.9.23.
doi: 10.3980/j.issn.1672-5123.2018.9.23 |
| 82 |
Zhang S, Zhang J, Xu XY, et al. [The efficacy of intravitreal injection of tissue plasminogen activator, ranibizumab and C3F8 in the treatment of early submacular hemorrhage induce to polypoid choroid vasculopathy]. Chin J Ocul Fundus Dis 2018 ; 34(5):448-52. Chinese. doi: 10.3760/cma.j.issn.1005-1015.2018.05.007.
doi: 10.3760/cma.j.issn.1005-1015.2018.05.007 |
| 83 |
Yan J, Wu JH. [Effects of photodynamic therapy with Ranibizumab on visual acuity and postoperative complications in patients with PCV complicated with vitreous hemorrhage]. Int Eye Sci 2018 ; 18(12):2209-12. Chinese. doi: 10.3980/j.issn.1672-5123.2018.12.20.
doi: 10.3980/j.issn.1672-5123.2018.12.20 |
| 84 |
Kimura S, Morizane Y, Hosokawa M, et al. Submacular hemorrhage in polypoidal choroidal vasculopathy treated by vitrectomy and subretinal tissue plasminogen activator. Am J Ophthalmol 2015 ; 159(4):683-9. doi: 10.1016/j.ajo.2014.12.020.
doi: 10.1016/j.ajo.2014.12.020 |
| 85 |
Kim JH, Chang YS, Lee DW, et al. Quantification of retinal changes after resolution of submacular hemorrhage secondary to polypoidal choroidal vasculopathy. Jpn J Ophthalmol 2018 ; 62(1):54-62. doi: 10.1007/s10384-017-0549-2.
doi: 10.1007/s10384-017-0549-2 |
| 86 |
Cheng Y, Shi X, Qu JF, et al. Comparison of the 1-year outcomes of conbercept therapy between two different angiographic subtypes of polypoidal choroidal vasculopathy. Chin Med J (Engl) 2016 ; 129(21):2610-16. doi: 10.4103/0366-6999.192779.
doi: 10.4103/0366-6999.192779 |
| 87 |
Ye LH, Cai Y, Shi X, et al. One-year results of intravitreal conbercept in treatment-naïve subjects with polypoidal choroidal vasculopathy. Graefes Arch Clin Exp Ophthalmol 2021 ; 259(6):1455-62. doi: 10.1007/s00417-020-04988-y.
doi: 10.1007/s00417-020-04988-y |
| 88 |
Qi HJ, Jin EZ, Zhao MW. One-year outcomes of intravitreal conbercept combined rescue therapy for polypoidal choroidal vasculopathy in a Chinese population: a real-life clinical data. Int J Ophthalmol 2019 ; 12(1):51-7. doi: 10.18240/ijo.2019.01.08.
doi: 10.18240/ijo.2019.01.08 |
| 89 |
Peng Y, Zhang X, Li M, et al. Short-term efficacy of intravitreal conbercept in treatment-naive patients with polypoidal choroidal vasculopathy. Drug Des Devel Ther 2018; 12:339-45. doi: 10.2147/dddt.S158368.
doi: 10.2147/dddt.S158368 |
| 90 |
Li F, Ma A, Zhao B. Comparison of the efficacy of three loading doses of intravitreal injection of conbercept with injection combined with PDT for the treatment of PCV. Biomed Res Int 2020; 2020:2428348. doi: 10.1155/2020/2428348.
doi: 10.1155/2020/2428348 |
| [1] | 刘勇, 姚思远, 周熹, 刘书中, 边焱焱. 便秘与胸椎肿瘤患者下肢肌力下降的相关性:单中心回顾性横断面研究[J]. Chinese Medical Sciences Journal, 2023, 38(2): 109-116. |
| [2] | 田丽娟, 姚允泰, 袁素, 戴峥. 右美托咪定对老年患者围术期血流动力学稳定性影响的系统评价与Meta分析[J]. Chinese Medical Sciences Journal, 2023, 38(1): 1-10. |
| [3] | 姚允泰, 袁昕, 何丽仙, 于宜平, 杜雨, 刘刚, 田丽娟, 马祖轩, 张永保, 马洁. 患者血液管理:阜外医院单中心证据和实践[J]. Chinese Medical Sciences Journal, 2022, 37(3): 246-260. |
| [4] | 张扬, 卞爱玲, 梁安怡, 莫菲, 程钢炜. 二氧化碳激光辅助深层巩膜切除术联合小梁切除术治疗1例继发于Weill-Marchesani综合征的难治性儿童青光眼[J]. Chinese Medical Sciences Journal, 2022, 37(2): 159-163. |
| [5] | 陈小坤, 苗齐, 朱铁楠, 张超纪. 心脏手术中血友病患者的替代疗法:3例报告[J]. Chinese Medical Sciences Journal, 2022, 37(1): 79-81. |
| [6] | 姚允泰, 何丽仙, 李丽萍. 阜外医院麻醉管理:实践、证据和结局[J]. Chinese Medical Sciences Journal, 2021, 36(3): 234-251. |
| [7] | 马璐璐,黄宇光. 胸腔镜胸腺肿瘤切除术膈神经损伤导致低氧血症:2例病例报告和文献综述[J]. Chinese Medical Sciences Journal, 2020, 35(2): 191-194. |
| [8] | 高原,谭珂,孙坚,姜涛,邹晓文. 混合现实技术在医学手术可视化中的应用研究[J]. Chinese Medical Sciences Journal, 2019, 34(2): 103-109. |
| [9] | 白冰, 田园, 张越伦, 马满娇, 虞雪融, 黄宇光. 后路脊柱手术隐性失血的预测[J]. Chinese Medical Sciences Journal, 2019, 34(1): 38-44. |
| [10] | 张雪, 虞雪融, 黄宇光. 神经外科手术病人快速血栓弹力图指标与术中出血量关系[J]. Chinese Medical Sciences Journal, 2017, 32(2): 69-74. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
|