Chinese Medical Sciences Journal ›› 2023, Vol. 38 ›› Issue (2): 130-137.doi: 10.24920/004183
• Original Article • Previous Articles Next Articles
Primary Ovarian Small Cell Carcinoma of Pulmonary Type: Analysis of 6 Cases and Review of 31 Cases in the Literatures
Xu Chen1, Hong-Ling Liu2, Jin-Sui Wang1, Feng-Hui Zhao1, *( )
)
- 1Department of Pathology, Gansu Provincial Hospital, Lanzhou 730000, China
2Department of Occupational Medicine, Third Gansu Provincial Hospital, Lanzhou 730000, China
-
Received:2022-10-18Accepted:2023-04-28Published:2023-06-30Online:2023-06-08 -
Contact:*zhaofh0931@163.com
| Primary small cell ovarian cancer of pulmonary type (SCCOPT) is a rare ovarian tumor with a poor prognosis. This study retrospectively summarized the clinical, imaging, laboratory, and pathological characteristics of 6 SCCOPT cases who were pathologically diagnosed at the Gansu Provincial Hospital and of 31 cases reported in literatures. The result revealed that the SCCOPT of all patients showed positive expression of sex-determining region of Y chromosome-related high-mobility-group box 2 (SOX-2), which indicating that SOX-2 could be a biomarker used to diagnose SCCOPT. |
Cite this article
Xu Chen, Hong-Ling Liu, Jin-Sui Wang, Feng-Hui Zhao. Primary Ovarian Small Cell Carcinoma of Pulmonary Type: Analysis of 6 Cases and Review of 31 Cases in the Literatures[J].Chinese Medical Sciences Journal, 2023, 38(2): 130-137.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
Table 1.
Clinical features and treatment methods of SCCOPT"
| References | Age (yrs) | Clinical symptoms | Tumor staging | Tumor diameter (cm) | Operation | Chemotherapy | Overall survival time (months) |
|---|---|---|---|---|---|---|---|
| Eichhorn et al.[ | 62 | pelvic mass | ⅠA (no metastasis ) | 21.5 | RSO | NR | 4, die from disease |
| Eichhorn et al.[ | 59 | pelvic mass | ⅠA (no metastasis) | 17.0 | TAH, RSO | NR | NR |
| Eichhorn et al.[ | 55 | pelvic mass | ⅠA (no metastasis) | 26.0 | TAH, BSO | NR | NR |
| Eichhorn et al.[ | 30 | abdominal mass | ⅠC (no metastasis) | 20.0 | BSO | NR | 6, die from disease |
| Eichhorn et al.[ | 84 | abdominal mass | ⅡB (peritoneal metastasis) | 13.5 | BSO | NR | 1, die from disease |
| Eichhorn et al.[ | 76 | vaginal bleeding and pelvic mass | ⅢB (peritoneal metastasis) | NR | TAH, BSO | NR | 12, die from disease |
| Eichhorn et al.[ | 50 | vaginal bleeding and abdominal mass | ⅢB (peritoneal metastasis) | NR | TAH, BSO | NR | NR |
| Eichhorn et al.[ | 72 | pelvic mass | ⅢB (peritoneal metastasis) | 4.5 | TAH, BSO | cisplatin, cyclophosphamide | NR |
| Eichhorn et al.[ | 64 | pelvic mass | ⅢB (peritoneal metastasis) | 5.5 | TAH, BSO | cisplatin, cyclophosphamide | 12, die from disease |
| Eichhorn et al.[ | 49 | pelvic mass and ascites | ⅢB (peritoneal metastasis) | 16.0 | LSO | cisplatin, cyclophosphamid, doxorubicin | 8, die from disease |
| Eichhorn et al.[ | 46 | abdominal pain and mass | ⅢC (peritoneal metastasis) | 9.5 | TAH, BSO | cisplatin, cyclophosphamide, etoposide | 13, die from disease |
| Chang et al.[ | 22 | pelvic mass | ⅢA (peritoneal metastasis ) | 18.0 | TAH, LSO | cisplatin, cyclophosphamide | 19, die from disease |
| Fukunaga et al.[ | 60 | abdominal mass | Ⅳ (lung metastasis) | 14.0 | TAH, RSO | cisplatin, cyclophosphamide | 10, die from lung metastasis |
| Lim et al.[ | 28 | vaginal bleeding, pelvic mass, and acute abdomen | ⅢB (peritoneal metastasis ) | 20.0 | TAH, RSO | NR | NR |
| Mebis et al.[ | 54 | pelvic mass | ⅢA (peritoneal metastasis | 12.0 | TAH, BSO | cisplatin, etoposide | 22, die from disease |
| Wang et al.[ | 22 | abdominal mass and ascites | IV (extrapelvic nodal metastasis) | 4.3 | TAH, BSO, LYM, OMT | NR | NR |
| Rund et al.[ | 56 | vaginal bleeding and ascites | Ⅳ (liver metastasis) | 12.0 | TAH, RSO | cisplatin, etoposide | 7, die from brain metastasis |
| Rund et al.[ | 39 | pelvic mass | ⅢB (peritoneal metastasis) | 15.0 | TAH, BSO | Cisplatin, etoposide, taxol | 16, die from disease |
| Marie et al.[ | 32 | abdominal mass and ascites | Ⅳ (lung metastasis) | 20.0 | TAH, BSO | paclitaxel, carbiplatin | 15, die from lung metastasis |
| Suzuki et al.[ | 49 | pelvic mass | Ⅳ (liver metastasis) | 15.0 | TAH, BSO | paclitaxel, carbiplatin | 25, die from lung metastasis |
| Tsolakidis et al.[ | 55 | abdominal mass and pain | Ⅳ (liver metastasis) | 8.0 | TAH, BSO, LYM | carboplatin, etoposide | 21, die from brain metastasis |
| Ikota et al.[ | 65 | abdominal mass and pain | Ⅳ (liver metastasis) | 18.0 | NR | NR | 6, die from brain metastasis |
| Kurasaki et al.[ | 54 | pelvic mass | ⅢA (peritoneal metastasis) | 14.0 | TAH, LSO, OMT | paclitaxel | 22, die from disease |
| Oyang et al.[ | 56 | abdominal mass and pain | Ⅳ (brain metastasis) | 5.0 | TAH, BSO, LYM, OMT | cispatin, etoposide, | NR |
| Rubio et al.[ | 80 | pelvic mass | Ⅳ (liver metastasis) | 20.0 | TAH, BSO | cisplatin, etoposide, paclitaxel | 21, die from lung metastasis |
| Hashimoto et al.[ | 75 | abdominal pain and ascites | Ⅳ (lung metastasis) | 12.5 | TAH, BSO, OMT | cisplatin, etoposide | NR |
| Kalampokas et al.[ | 77 | pelvic mass | ⅡB (peritoneal metastasis) | 11.3 | TAH, BSO | cyclophosphamide, doxorubicin | NR |
| Oneda et al.[ | 72 | abdominal mass and vaginal bleeding | ⅢC (peritoneal metastasis) | 6.0 | TAH, LSO, LYM | carboplatin | 10, die from disease |
| Ishikawa et al.[ | 50 | pelvic mass and ascites | Ⅳ (omentum and extrapelvic nodal metastasis) | 12.0 | TAH, LSO, OMT | cisplatin, etoposide, paclitaxel | 12, die from lung metastasis |
| Li et al.[ | 45 | abdominal pain and mass | Ⅳ (bone metastasis) | 10.0 | TAH, BSO, OMT, LYM | paclitaxel, etoposide | NR |
| Asom et al.[ | 80 | abdominal mass, ascites, and vaginal bleeding | Ⅳ (omentum and extrapelvic nodal metastasis) | 18.5 | TAH, RSO, LYM | carboplatin treatment after radiotherapy | 16, die from lung metastasis |
| Case 1 | 65 | pelvic mass and lower abdominal pain | ⅢA (peritoneal metastasis) | 10.7 | TAH, BSO | cisplatin, doxorubicin | 27, die from disease |
| Case 2 | 42 | abdominal mass and ascites | Ⅳ (liver metastasis) | 13.6 | TAH, BSO, OMT, LYM, AP | cisplatin, cyclophosphamide | 10, die from brain metastasis |
| Case 3 | 53 | abdominal mass and lower abdominal pain | ⅢB (peritoneal metastasis) | 11.2 | TAH, BSO, OMT, LYM | cisplatin, paclictaxel | 16, die from disease |
| Case 4 | 79 | pelvic mass and vaginal bleeding | ⅢB (peritoneal metastasis) | 13.4 | TAH, BSO, OMT | carboplatin, paclictaxel | 24, die from disease |
| Case 5 | 61 | pelvic mass and ascites | Ⅵ (omentum and lung metastasis) | 15.8 | TAH, BSO, OMT, LYM, AP | carboplatin, etoposide | 9, die from lung metastasis |
| Case 6 | 73 | abdominal mass and lower abdominal pain | ⅡA (fallopian tube involvement) | 9.1 | TAH, BSO | cisplatin, paclictaxel | NA |
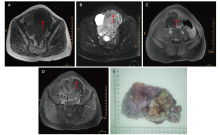
Figure 2.
Pelvic MRI findings and macroscopic pathology of a 42-year-old woman with SCCOPT (Case 2). (A) The lesion shows a low signal on a T1-weighted image (red arrow). (B) The lesion shows heterogeneously high intensity on T2-weighted image (red arrow). (C, D) The lesion shows contrast enhancement on enhanced MRI scan (red arrows). (E) The lesion appears as a complex cystic and solid mass with a papillary structure."

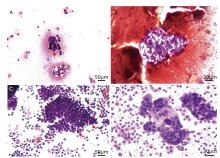
Figure 3.
Cytological findings of peritoneal effusion with smear test in two patients with SCCOPT. HE stain (A, B) A large number of atypical cells (red arrow) are aggregated together in the peritoneal effusion of Case 2. (C) A mass of tumor cells of Case 5 form some small clusters (red arrows). (D) The tumor cells in peritoneal effusion of Case 5 have a high nucleus-to-cytoplasm ratio with oval-to-round nuclei, fine granular chromatin, scant cytoplasm, and indistinct nucleoli."
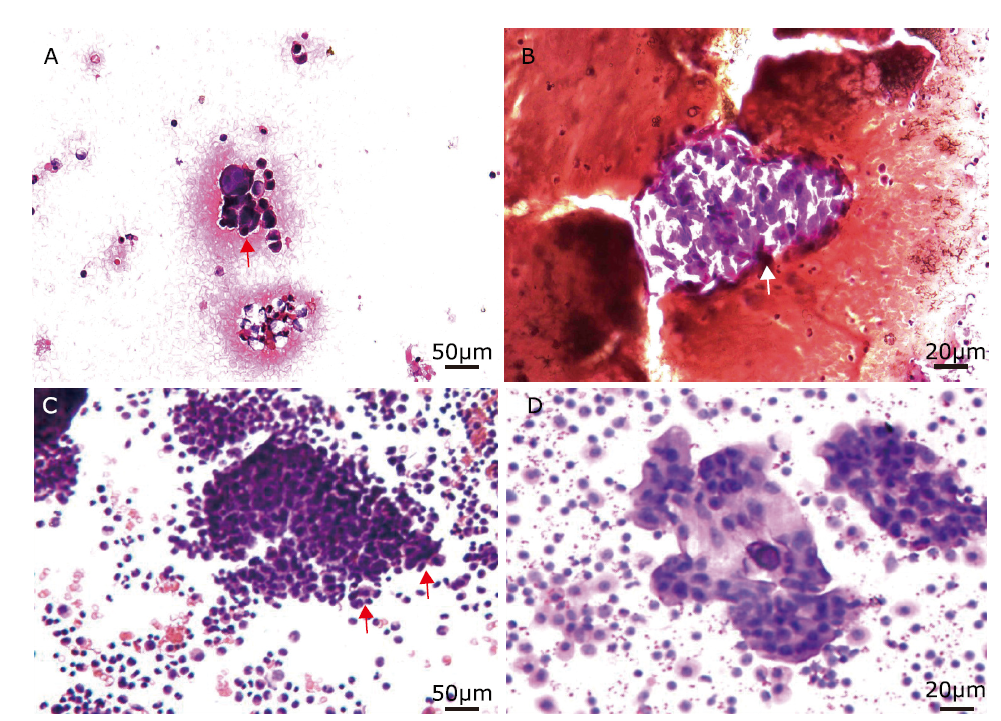
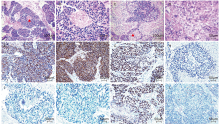
Figure 4.
Histological and immunohistochemical results in three patients with SCCOPT. (A, B) HE stains showing the tumor of Case 3 consisted of small cells with scant cytoplasm and mitotic nuclei, and local necrosis (red triangle). (C, D) SCCOPT of Case 6 coexisted with Brenner tumor (red triangle) (HE stain). (E) SCCOPT of Case 4 was positive for wide-spectrum (pan) cytokeratin (DAB stain). (F) SCCOPT of Case 4 was positive for CD56 (DAB stain). (G) SCCOPT of Case 4 was positive for synaptophysin (DAB stain). (H) SCCOPT of Case 4 was negative for chromogranin A (DAB stain). (I) SCCOPT of Case 4 was negative for neuron-specific enolase (DAB stain). (J) Case 4 was negative for thyroid transcription factor-1 (DAB stain). (K) SCCOPT of Case 4 was positive for sex-determining region of Y chromosome-related high-mobility-group box 2 (SOX-2, DAB stain). (L) SCCOPT of Case 4 was negative for somatostatin receptor type 2 (DAB stain)."

| 1 |
Vora M, Lacour RA, Black DR, et al. Neuroendocrine tumors in the ovary: histogenesis, pathologic differentiation, and clinical presentation. Arch Gynecol Obstet 2016 ; 293(3):659-5. doi: 10.1007/s00404-015-3865-0.
doi: 10.1007/s00404-015-3865-0 |
| 2 |
De Leo A, Santini D, Ceccarelli C, et al. What is new on ovarian carcinoma: integrated morphologic and molecular analysis following the New 2020 World Health Organization Classification of Female Genital Tumors. Diagnostics 2021 ; 11(4): 697. doi: 10.3390/diagnostics11040697.
doi: 10.3390/diagnostics11040697 |
| 3 |
Eichhorn JH, Young RH, Scully RE. Primary ovarian small cell carcinoma of pulmonary type. A clinicopathologic, immunohistologic, and flow cytometric analysis of 11 cases. Am J Surg Pathol 1992 ; 16(10):926-38. doi: 10.1097/00000478-199210000-00002.
doi: 10.1097/00000478-199210000-00002 |
| 4 |
Chang DHC, Hsueh S, Soong YK. Small cell carcinoma with neurosecretory granules arising in an ovarian dermoid cyst. Gynecol Oncol 1992 ; 46(2):246-50. doi: 10.1097/00000478-199210000-00002
doi: 10.1097/00000478-199210000-00002 |
| 5 |
Fukunaga M, Endo Y, Miyazawa Y, et al. Small cell neuroendocrine carcinoma of the ovary. Virchows Archiv 1997 ; 430(4):343-8. doi: 10.1007/BF01092759.
doi: 10.1007/BF01092759 |
| 6 |
Lim SC, Choi SJ, Suh CH. A case of small cell carcinoma arising in a mature cystic teratoma of the ovary. Pathol Int 1998 ; 48(10):834-9. doi: 10.1111/j.1440-1827.1998.tb03846.x.
doi: 10.1111/j.1440-1827.1998.tb03846.x |
| 7 | Mebis J, De Raeve H, Baekelandt M, et al. Primary ovarian small cell carcinoma of the pulmonary type: a case report and review of the literature. Eur J Gynecol Oncol 2004 ; 25(2):239-41. |
| 8 |
Grandjean M, Legrand L, Waterkeyn M, et al. Small cell carcinoma of pulmonary type inside a microinvasive mucinous cystadenocarcinoma of the ovary: a case report. Int J Gynecol Pathol 2007 ; 26(4):426-31. doi: 10.1097/pgp.0b013e318039373a.
doi: 10.1097/pgp.0b013e318039373a |
| 9 |
Ishikawa A, Fujisawa H, Yasumura N, et al. Ovarian pulmonary-type small cell carcinoma diagnosed by means of ascitic fluid cytology: a case report with immunocytochemical analysis. Cytopathology 2021 ; 32(2):250-2. doi: 10.1111/cyt.12918.
doi: 10.1111/cyt.12918 |
| 10 |
Rund CR, Fischer EG. Perinuclear dot-like cytokeratin 20 staining in small cell neuroendocrine carcinoma of the ovary (pulmonary-type). Appl Immunohistochem Mol Morphol 2006 ; 14(2):244-8. doi: 10.1097/01.pai.0000155796.49278.62.
doi: 10.1097/01.pai.0000155796.49278.62 |
| 11 |
Rubio A, Schuldt M, Chamorro C, et al. Ovarian small cell carcinoma of pulmonary type arising in mature cystic teratomas with metastases to the contralateral ovary. Int J Surg Pathol 2015 ; 23(5):388-92. doi: 10.1177/1066896915586254.
doi: 10.1177/1066896915586254 |
| 12 |
Suzuki N, Kameyama K, Hirao T, et al. A case of pulmonary type of ovarian small cell carcinoma. J Obstet Gynecol Res 2007 ; 33(2):203-6.doi: 10.1111/j.1447-0756.2007.00494.x.
doi: 10.1111/j.1447-0756.2007.00494.x |
| 13 | Tsolakidis D, Papanikolaou A, Ktenidis K, et al. Primary ovarian small cell carcinoma of pulmonary type with enlarged paraaortic lymph node masses: a case report and review of the literature. Eur J Gynecol Oncol 2012 ; 33(3):312-5. |
| 14 |
Kurasaki A, Sakurai N, Yamamoto Y, et al. Ovarian pulmonary-type small cell carcinoma: case report and review of the literature. Int J Gynecol Pathol 2013 ; 32(5): 464-70. doi: 10.1097/PGP.0b013e31826d7ea8.
doi: 10.1097/PGP.0b013e31826d7ea8 |
| 15 |
Ikota H, Kaneko K, Takahashi S, et al. Malignant transformation of ovarian mature cystic teratoma with a predominant pulmonary type small cell carcinoma component. Pathol Int 2012 ; 62(4):276-80. doi: 10.1111/j.1440-1827.2012.02804.x.
doi: 10.1111/j.1440-1827.2012.02804.x |
| 16 |
Hashimoto H, Kurata A, Fujita K, et al. Ovarian small cell carcinoma of pulmonary type appearing in ante-mortem ascites: an autopsy case and review of the literature. Human Pathol: Case Rep 2015 ; 2(3):67-72. doi: 10.1016/j.ehpc.2015.02.003.
doi: 10.1016/j.ehpc.2015.02.003 |
| 17 |
Kalampokas E, Sharma V, Gagliardi T, et al. Primary small cell ovarian cancer of pulmonary type: a case report. Gynecol Oncol Rep 2018; 24:10-3. doi: 10.1016/j.gore.2018.02.003.
doi: 10.1016/j.gore.2018.02.003 |
| 18 |
Oneda E, Zorzi F, Gorio A, et al. Differential diagnosis of small cell carcinoma of the ovary or ovarian metastases of small cell carcinoma of the lung: a case report and review of the literature. Case Rep Oncol 2020 ; 13(2):822-8. doi: 10.1159/000507978.
doi: 10.1159/000507978 |
| 19 |
Asom AS, Lastra RR, Hasan Y, et al. Small cell carcinoma of the ovary, pulmonary type: a role for adjuvant radiotherapy after carboplatin and etoposide? Gynecol Oncol Rep 2022; 39:100925. doi: 10.1016/j.gore.2022.100925.
doi: 10.1016/j.gore.2022.100925 |
| 20 |
Wang YL, Zou SY, Yue XH, et al. Small cell carcinoma of the ovary, pulmonary type: a case report and literature review. J Clin Ex Pathol 2005(3):367-8. doi: 10.13315/j.cnki.cjcep.2005.03.041.
doi: 10.13315/j.cnki.cjcep.2005.03.041 |
| 21 |
Ouyang ZB, Su HH, Zhong BL, et al. Primary ovarian small cell carcinoma of pulmonary type: a case report and literature review. Chin J Cancer Prev Treat 2014 ; 21(1):69-71. doi: 10.16073/j.cnki.cjcpt.2014.01.001.
doi: 10.16073/j.cnki.cjcpt.2014.01.001 |
| 22 |
Li Y, Yang GQ, Shuang YJ, et al. Primary ovarian small cell carcinoma of pulmonary type: a case report and literature review. Diagnos Imaging Intervent Radiol 2021 ; 30(1):64-7. doi: 10.3969/j.issn.1005-8001.2021.01.013.
doi: 10.3969/j.issn.1005-8001.2021.01.013 |
| 23 |
Shahrokhi P, Emami-Ardekani A, Karamzade-Ziarati N. SSTR-based theranostics in neuroendocrine prostate cancer (NEPC). Clin Transl Imaging 2022 ; 22(11):1-8. doi: 10.1007/s40336-022-00535-3.
doi: 10.1007/s40336-022-00535-3 |
| 24 |
Burki TK. Small-cell carcinoma of the ovary, hypercalcaemic type. Lancet Oncol 2014; 15(6):e201. doi: 10.1016/S1470-2045(14)70144-5.
doi: 10.1016/S1470-2045(14)70144-5 |
| 25 |
Errichiello E, Mustafa N, Vetro A, et al. SMARCA4 inactivating mutations cause concomitant Coffin-Siris syndrome, microphthalmia and small-cell carcinoma of the ovary hypercalcaemic type. J Pathol 2017 ; 243(1):9-15. doi: 10.1002/path.4926.
doi: 10.1002/path.4926 |
| 26 |
Gandhi JS, Alnoor F, Sadiq Q, et al. SMARCA4 (BRG1) and SMARCB1 (INI1) expression in TTF-1 negative neuroendocrine carcinomas including merkel cell carcinoma. Pathol Res Pract 2021; 219:153341. doi: 10.1016/j.prp.2021.153341.
doi: 10.1016/j.prp.2021.153341 |
| 27 |
William WN, Glisson BS. Novel strategies for the treatment of small-cell lung carcinoma. Nat Rev Clin Oncolo 2011 ; 8(10):611-9. doi: 10.1038/nrclinonc.2011.90.
doi: 10.1038/nrclinonc.2011.90 |
| [1] | Shu-bo Tian, Jian-chun Yu*, Wei-ming Kang, Zhi-qiang Ma, Xin Ye, Chao Yan, Ya-kai Huang. Effect of Neoadjuvant Chemotherapy Treatment on Prognosis of Patients with Advanced Gastric Cancer: a Retrospective Study [J]. Chinese Medical Sciences Journal, 2015, 30(2): 84-89. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
|



