Chinese Medical Sciences Journal ›› 2021, Vol. 36 ›› Issue (2): 150-157.doi: 10.24920/003850
• Case Reports • Previous Articles Next Articles
Alternating Hemiplegia of Childhood Caused by ATP1A3 Mutations: A Report of Two Cases
Guange Yang( ), Zhongli Zhao, Yang Yang, Li Lin, Conglei Song, Xiaocui Wang, Bin Yang
), Zhongli Zhao, Yang Yang, Li Lin, Conglei Song, Xiaocui Wang, Bin Yang
- Medical Department of Neurology, Anhui Provincial Children’s Hospital, Hefei 230053, China
-
Received:2021-02-10Published:2021-06-30Online:2021-05-31 -
Contact:Guange Yang E-mail:guangeyang@163.com
| The authors reported two boys with alternating hemiplegia who carried heterozygous missense mutations in the ATP1A3 gene (OMIM: 614820): c.3025 (exon 22) A>G (p.K1009E) and c.2443 (exon 18) G>A (p.E815K), a primary pathogenic gene for alternating hemiplegia of childhood. |
Cite this article
Guange Yang, Zhongli Zhao, Yang Yang, Li Lin, Conglei Song, Xiaocui Wang, Bin Yang. Alternating Hemiplegia of Childhood Caused by ATP1A3 Mutations: A Report of Two Cases[J].Chinese Medical Sciences Journal, 2021, 36(2): 150-157.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
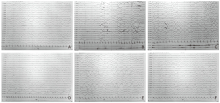
Figure 1.
Electroencephalogram (EEG) changes of the two patients: A. Slow background in case 1. B. There is a focal episode in the wake-up phase, as characterized by binocular gaze and lip smacking, with a concomitant EEG in the left temporal region—high slow waves, mixed with sharp and spine waves in case 1. C. Discharge in the left temporal region involved the ipsilateral frontal region in case 1. D. Slow background in case 2. E, F. Asynchronous emission of slow and tip slow waves in bilateral occipital and posterior temporal regions during sleep in case 2."


Figure 2.
Cranial MRI of case 2 showing hyperintense patchy signals of the left parahippocampal gyrus on diffusion-weighted imaging (arrows, C, D) and apparent diffusion coefficent (E), but normal signals on T1 weighted imaging (A), T2 weighted imaging (B) and magnetic resonance angiography (F)."

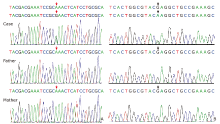
Figure 3.
DNA sequencing results of ATP1A3 mutations in the two cases based on first-generation sequencing. A. Case 1, heterozygous missense mutation of ATP1A3, c.3025 (exon 22) A>G. B. Case 2, heterozygous missense mutation ofATP1A3, c.2443 (exon 18) G>A. No mutation was found at these sites in the parents. "
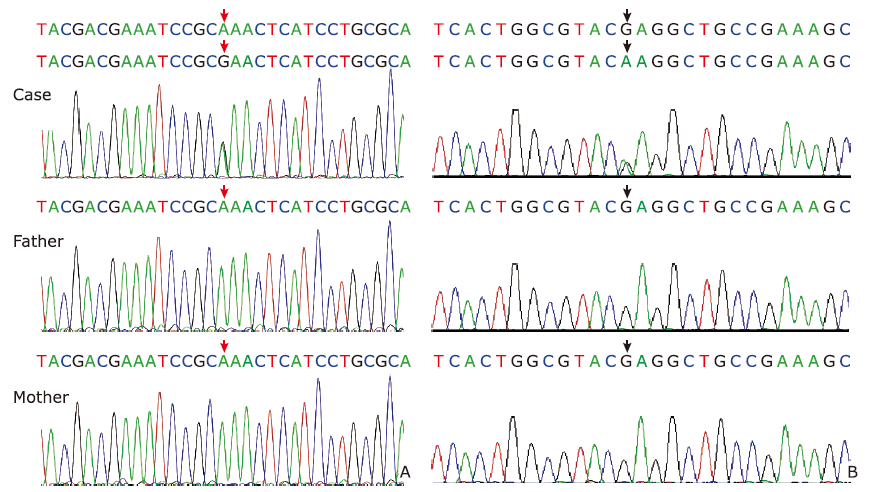
Table 1
Mutations in the ATP1A3 gene in two pediatric patients "
| No. | Mutation | Report | Amino acid change | Source of mutation | Rs number | 1000 Genomes/GnomAD/ESP | SIFT/PolyPhen | GERP | ACMG guideline |
|---|---|---|---|---|---|---|---|---|---|
| Case 1 | c.3025A>G | Not | p.K1009E(NM _001256214) | de novo | None | NA/NA/NA | Damaging (0.001)/possibly damaging (0.651) | D (3.32) | Likely pathogenic (PS2+PM1+PM2+PP3) |
| Case 2 | c.2443G>A | Yes | p.E815K (NM_152296) | de novo | rs387907281 | NA/NA/NA | Damaging (0.001)/porobably damaging (1.0) | D (3.69) | Pathogenic (PS2+PS1+PM+PM1+PM2+PM+PP5) |
| [1.] |
Verret S, Steele JC. Alternating hemiplegia in childhood: a report of eight patients with complicated migraine beginning in infancy. Pediatrics 1971; 47(4):675-80.
pmid: 5089756 |
| [2.] |
Neville BGR, Ninan M. The treatment and management of alternating hemiplegia of childhood. Dev Med Child Neurol 2007; 49(10):777-80. doi: 10.1111/j.1469-8749.2007.00777.x.
doi: 10.1111/j.1469-8749.2007.00777.x |
| [3.] |
Bourgeois M, Aicardi J, Goutieres F. Alternating hemiplegia of childhood. J Pediatr 1993; 122(5 Pt 1):673-9. doi: 10.1016/s0022-3476(06)80003-x.
doi: 10.1016/s0022-3476(06)80003-x |
| [4.] |
Heinzen EL, Swoboda KJ, Hitomi Y, et al. De novo mutations in ATP1A3 cause alternating hemiplegia of childhood . Nat Genet 2012; 44(9):1030-4. doi: 10.1038/ng.2358.
doi: 10.1038/ng.2358 |
| [5.] |
Rosewich H, Thiele H, Ohlenbusch A, et al. Heterozygous denovo mutations in ATP1A3 in patients with alternating hemiplegia of childhood: a whole-exome sequencing gene-identification study . Lancet Neurol 2012; 11(9):764-73. doi: 10.1016/S1474-4422(12)70182-5.
doi: 10.1016/S1474-4422(12)70182-5 pmid: 22850527 |
| [6.] |
Capuano A, Garone G, Tiralongo G, et al. Alternating hemiplegia of childhood: understanding the genotypephenotype relationship of ATP1A3 variations . Appl Clin Genet 2020; 13:71-81. doi: 10.2147/TACG.S210325.
doi: 10.2147/TACG.S210325 |
| [7.] |
Yang X, Gao H, Zhang J, et al. ATP1A3 mutations and genotype-phenotype correlation of alternating hemiplegia of childhood in Chinese patients . PLoS One 2014; 9(5):e97274. doi: 10.1371/journal.pone.0097274.
doi: 10.1371/journal.pone.0097274 |
| [8.] |
Pisciotta L, Gherzi M, Stagnaro M, et al. Alternating hemiplegia of childhood: pharmacological treatment of 30 Italian patients. Brain Dev 2017; 39(6):521-8. doi: 10.1016/j.braindev.2017.02.001.
doi: 10.1016/j.braindev.2017.02.001 |
| [9.] |
Sasaki M, Ishii A, Saito Y, et al. Genotype-phenotype correlations in alternating hemiplegia of childhood. Neurology 2014; 82(6):482-90. doi: 10.1212/WNL.0000000000000102.
doi: 10.1212/WNL.0000000000000102 |
| [10.] |
Uchitel J, Helseth A, Prange L, et al. The epileptology of alternating hemiplegia of childhood. Neurology 2019; 93(13):e1248-e59. doi: 10.1212/WNL.0000000000008159.
doi: 10.1212/WNL.0000000000008159 |
| [11.] |
Sasaki M, Ishii A, Saito Y, et al. Progressive brain atrophy in alternating hemiplegia of childhood. Mov Disord Clin Pract 2017; 4(3):406-11. doi: 10.1002/mdc3.12451.
doi: 10.1002/mdc3.12451 |
| [12.] |
Jaffer F, Avbersek A, Vavassori R, et al. Faulty cardiac repolarization reserve in alternating hemiplegia of childhood broadens the phenotype. Brain 2015; 138(Pt 10):2859-74. doi: 10.1093/brain/awv243.
doi: 10.1093/brain/awv243 |
| [13.] |
Marzin P, Mignot C, Dorison N, et al. Early-onset encephalopathy with episodic movement disorders and epileptic seizures without hemiplegic attacks: about three children with novel ATP1A3 mutations. Brain Dev 2018; 40(9):768-74. doi: 10.1016/j.braindev.
doi: 10.1016/j.braindev |
| [14.] |
Torres A, Brownstein CA, Tembulkar SK, et al. De novo ATP1A3 and compound heterozygous NLRP3 mutations in a child with autism spectrum disorder, episodic fatigue and somnolence, and muckle-wells syndrome . Mol Genet Metab Rep 2018; 16:23-9. doi: 10.1016/j.ymgmr.2018.06.001.
doi: 10.1016/j.ymgmr.2018.06.001 pmid: 29922587 |
| [15.] |
Smedemark-Margulies N, Brownstein CA, Vargas S, et al. A novel de novo mutation in ATP1A3 and childhood-onset schizophrenia . Cold Spring Harb Mol Case Stud 2016; 2(5):a001008. doi: 10.1101/mcs.a001008.
doi: 10.1101/mcs.a001008 |
| [16.] |
Chaumette B, Ferrafiat V, Ambalavanan A, et al. Missense variants in ATP1A3 and FXYD gene family are associated with childhood-onset schizophrenia . Mol Psychiatry 2020; 25(4):821-30. doi: 10.1038/s41380-018-0103-8.
doi: 10.1038/s41380-018-0103-8 |
| [17.] |
Panagiotakaki E, De Grandis E, Stagnaro M, et al. Clinical profile of patients with ATP1A3 mutations in alternating hemiplegia of childhood—a study of 155 patients . Orphanet J Rare Dis 2015; 10:123. doi: 10.1186/s13023-015-0335-5.
doi: 10.1186/s13023-015-0335-5 pmid: 26410222 |
| [18.] |
Paciorkowski AR, McDaniel SS, Jansen LA, et al. Novel mutations in ATP1A3 associated with catastrophic early life epilepsy, episodic prolonged apnea, and postnatal microcephaly. Epilepsia 2015; 56(3):422-30. doi: 10.1111/epi.12914.
doi: 10.1111/epi.12914 pmid: 25656163 |
| [19.] |
Pavlidis E, Uldall P, Gøbel Madsen C, et al. Alternating hemiplegia of childhood and a pathogenic variant of ATP1A3: a case report and pathophysiological considerations . Epileptic Disord 2017; 19(2):226-30. doi: 10.1684/epd.2017.0913.
doi: 10.1684/epd.2017.0913 pmid: 28637637 |
| [20.] |
Jiang WJ, Chi ZF, Ma L, et al. Topiramate: a new agent for patients with alternating hemiplegia of childhood. Neuropediatrics 2006; 37(4):229-33. doi: 10.1055/s-2006-924721.
doi: 10.1055/s-2006-924721 pmid: 17177149 |
| [21.] |
Masoud M, Prange L, Wuchich J, et al. Diagnosis and treatment of alternating hemiplegia of childhood. Curr Treat Options Neurol 2017; 19(2):8. doi: 10.1007/s11940-017-0444-7.
doi: 10.1007/s11940-017-0444-7 |
| [22.] |
Ju J, Hirose S, Shi XY, et al. Treatment with oral ATP decreases alternating hemiplegia of childhood with de novo ATP1A3 mutation . Orphanet J Rare Dis 2016; 11(1):55. doi: 10.1186/s13023-016-0438-7.
doi: 10.1186/s13023-016-0438-7 |
| [23.] |
Dundar NO, Cavusoglu D, Kaplan YC, et al. An option to consider for alternating hemiplegia of childhood: aripiprazole. Clin Neuropharmacol 2019; 42(3):88-90. doi: 10.1097/WNF.0000000000000339.
doi: 10.1097/WNF.0000000000000339 pmid: 30893129 |
| [24.] |
Kramer PL, Mineta M, Klein C, et al. Rapid-onset dystonia-parkinsonism: linkage to chromosome 19q13. Ann Neurol 1999; 46(2):176-82. doi: 10.1002/1531-8249 (199908)46:2<176::aid-ana6>3.0.co;2-2.
doi: 10.1002/1531-8249 pmid: 10443882 |
| [25.] |
Haq IU, Snively BM, Sweadner KJ, et al. Revising rapid-onset dystonia-parkinsonism: broadening indications for ATP1A3 testing. Mov Disord 2019; 34(10):1528-36. doi: 10.1002/mds.27801.
doi: 10.1002/mds.27801 |
| [26.] |
Wilcox R, Brænne I, Brüggemann N, et al. Genome sequencing identifies a novel mutation in ATP1A3 in a family with dystonia in females only . J Neurol 2015; 262(1):187-93. doi: 10.1007/s00415-014-7547-9.
doi: 10.1007/s00415-014-7547-9 |
| [27.] |
Oblak AL, Hagen MC, Sweadner KJ, et al. Rapid-onset dystonia-parkinsonism associated with the I758S mutation of the ATP1A3 gene: a neuropathologic and neuroanatomical study of four siblings . Acta Neuropathol 2014; 128(1):81-98. doi: 10.1007/s00401-014-1279-x.
doi: 10.1007/s00401-014-1279-x |
| [28.] |
Yuan Y, Ran L, Lei L, et al. The expanding phenotypic spectrums associated with ATP1A3 mutation in a family with rapid-onset dystonia parkinsonism. Neurodegener Dis 2020; 20:84-9. doi: 10.1159/000511733.
doi: 10.1159/000511733 |
| [29.] |
Demos MK, van Karnebeek CD, Ross CJ, et al. A novel recurrent mutation in ATP1A3 causes CAPOS syndrome. Orphanet J Rare Dis 2014; 9:15. doi: 10.1186/1750-1172-9-15.
doi: 10.1186/1750-1172-9-15 |
| [30.] |
Heimer G, Sadaka Y, Israelian L, et al. CAOS—Episodic Cerebellar Ataxia, Areflexia, Optic Atrophy, and Sensorineural Hearing Loss: a third allelic disorder of theATP1A3 gene . J Child Neurol 2015; 30(13):1749-56. doi: 10.1177/0883073815579708.
doi: 10.1177/0883073815579708 |
| [31.] |
Dard R, Mignot C, Durr A, et al. Relapsing encephalopathy with cerebellar ataxia related to an ATP1A3 mutation . Dev Med Child Neurol 2015; 57(12):1183-6. doi: 10.1111/dmcn.12927.
doi: 10.1111/dmcn.12927 |
| [32.] |
Hully M, Ropars J, Hubert L, et al. Mosaicism in ATP1A3-related disorders: not just a theoretical risk. Neurogenetics 2017; 18(1):23-8. doi: 10.1007/s10048-016-0498-9.
doi: 10.1007/s10048-016-0498-9 |
| [33.] |
Yano ST, Silver K, Young R, et al. Fever-induced episodic weakness and encephalopathy, a new phenotype of ATP1A3 mutation. Pediatr Neurol 2017; 73:101-5. doi: 10.1016/j.pediatrneurol.2017.04.022.
doi: 10.1016/j.pediatrneurol.2017.04.022 |
| [34.] |
Nakamura Y, Hattori A, Nakashima M, et al. A de novo p. Arg756Cys mutation in ATP1A3 causes a distinct phenotype with prolonged weakness and encephalopathy triggered by fever . Brain Dev 2018; 40(3):222-5. doi: 10.1016/j.braindev.2017.09.010.
doi: 10.1016/j.braindev.2017.09.010 |
| [35.] |
Sabouraud P, Riquet A, Spitz MA, et al. Relapsing encephalopathy with cerebellar ataxia are caused by variants involving p. Arg756 in ATP1A3 . Eur J Paediatr Neurol 2019; 23(3):448-55. doi: 10.1016/j.ejpn.2019.02.004.
doi: 10.1016/j.ejpn.2019.02.004 |
| [36.] |
Rosewich H, Weise D, Ohlenbusch A, et al. Phenotypic overlap of alternating hemiplegia of childhood and CAPOS syndrome. Neurology 2014; 83(9):861-3. doi: 10.1212/WNL.0000000000000735.
doi: 10.1212/WNL.0000000000000735 pmid: 25056583 |
| [37.] |
Hayashida T, Saito Y, Ishii A, et al. Further characterization of CAPOS/CAOS syndrome with the Glu818 Lys mutation in the ATP1A3 gene: a case report . Brain Dev 2018; 40(7):576-81. doi: 10.1016/j.braindev.2018.03.004.
doi: 10.1016/j.braindev.2018.03.004 |
| [38.] |
Stenshorne I, Rasmussen M, Salvanos P, et al. Fever-related ataxia: a case report of CAPOS syndrome. Cerebellum Ataxias 2019; 6:2. doi: 10.1186/s40673-019-0096-3.
doi: 10.1186/s40673-019-0096-3 pmid: 31410291 |
| [39.] |
Schirinzi T, Graziola F, Nicita F, et al. Childhood rapid-onset ataxia: expanding the phenotypic spectrum of ATP1A3 mutations . Cerebellum 2018; 17(4):489-93. doi: 10.1007/s12311-018-0920-y.
doi: 10.1007/s12311-018-0920-y pmid: 29397530 |
| [1] | Kunfang Yang, Linyi Meng, Yuanfeng Zhang, Yongchen Yang, Hongyi Cheng, Zhihu Jiang, Hong Zhang, Yucai Chen. POLG Mutations Are Probably Rare in the Han Chinese Population [J]. Chinese Medical Sciences Journal, 2020, 35(4): 350-356. |
| [2] | Chao Wang, Chao You, Guo-qiang Han, Jun Wang, Yun-biao Xiong, Chuang-xi Liu. Electrocorticography-Guided Surgical Treatment of Solitary Supratentorial Cavernous Malformations with Secondary Epilepsy [J]. Chinese Medical Sciences Journal, 2014, 29(2): 112-116. |
| [3] | Xiao-liang Xing, Long-ze Sha, Yuan Yao, Yan Shen, Li-wen Wu*, and Qi Xu* . Spatio-temporal Expression Study of Phosphorylated 70-kDa Ribosomal S6 Kinase (p70S6k) in Mesial Temporal Lobe Epilepsy [J]. Chinese Medical Sciences Journal, 2012, 27(1): 7-10. |
| [4] | Resha Shrestha, Kuo Li, Wei Wang, Hai-ping Lian, and Mao-de Wang*. Electrocorticography with Direct Cortical Stimulation for a Left Temporal Glioma with Intractable Epilepsy [J]. Chinese Medical Sciences Journal, 2012, 27(1): 54-56. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
|

